Abstract
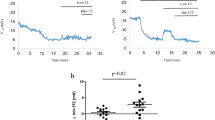
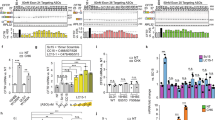
The potential for gene therapy to be an effective treatment for cystic fibrosis has been hampered by the limited gene transfer efficiency of current vectors. We have shown that recombinant Sendai virus (SeV) is highly efficient in mediating gene transfer to differentiated airway epithelial cells, because of its capacity to overcome the intra- and extracellular barriers known to limit gene delivery. Here, we have identified a novel method to allow the cystic fibrosis transmembrane conductance regulator (CFTR) cDNA sequence to be inserted within SeV (SeV-CFTR). Following in vitro transduction with SeV-CFTR, a chloride-selective current was observed using whole-cell and single-channel patch-clamp techniques. SeV-CFTR administration to the nasal epithelium of cystic fibrosis (CF) mice (CftrG551D and Cftrtm1UncTgN(FABPCFTR)#Jaw mice) led to partial correction of the CF chloride transport defect. In addition, when compared to a SeV control vector, a higher degree of inflammation and epithelial damage was found in the nasal epithelium of mice treated with SeV-CFTR. Second-generation transmission-incompetent F-deleted SeV-CFTR led to similar correction of the CF chloride transport defect in vivo as first-generation transmission-competent vectors. Further modifications to the vector or the host may make it easier to translate these studies into clinical trials of cystic fibrosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Johnson LG, Olsen JC, Sarkadi B, Moore KL, Swanstrom R, Boucher RC . Efficiency of gene transfer for restoration of normal airway epithelial function in cystic fibrosis. Nat Genet 1992; 2: 21–25.
Dorin JR, Farley R, Webb S, Smith SN, Farini E, Delaney SJ et al. A demonstration using mouse models that successful gene therapy for cystic fibrosis requires only partial gene correction. Gene Therapy 1996; 3: 797–801.
Ferrari S, Geddes DM, Alton EWFW . Barriers and new approaches for gene therapy and gene delivery in cystic fibrosis. Adv Drug Deliv Rev 2002; 54: 1373–1393.
Kitson C, Angel B, Judd D, Rothery S, Severs NJ, Dewar A et al. The extra- and intracellular barriers to lipid and adenovirus-mediated pulmonary gene transfer in native sheep airway epithelium. Gene Therapy 1999; 6: 534–546.
Ferrari S, Kitson C, Farley R, Steel R, Marriott C, Parkins DA et al. Mucus altering agents as adjuncts for nonviral gene transfer to airway epithelium. Gene Therapy 2001; 8: 1380–1386.
Wang G, Zabner J, Deering C, Launspach J, Shao J, Bodner M et al. Increasing epithelial junction permeability enhances gene transfer to airway epithelia in vivo. Am J Respir Cell Mol Biol 2000; 22: 129–138.
Chu Q, St George JA, Lukason M, Cheng SH, Scheule RK, Eastman SJ . EGTA enhancement of adenovirus-mediated gene transfer to mouse tracheal epithelium in vivo. Hum Gene Ther 2001; 12: 455–467.
Duan D, Yue Y, Yan Z, Yang Y, Engelhardt JF . Endosomal processing limits gene transfer to polarized airway epithelia by adeno-associated virus. J Clin Invest 2000; 105: 1573–1587.
Kobinger GP, Weiner DJ, Yu QC, Wilson JM . Filovirus-pseudotyped lentiviral vector can efficiently and stably transduce airway epithelia in vivo. Nat Biotechnol 2001; 19: 225–230.
Zhang L, Peeples ME, Boucher RC, Collins PL, Pickles RJ . Respiratory syncytial virus infection of human airway epithelial cells is polarized, specific to ciliated cells, and without obvious cytopathology. J Virol 2002; 76: 5654–5666.
Ziady AG, Kelley TJ, Milliken E, Ferkol T, Davis PB . Functional evidence of CFTR gene transfer in nasal epithelium of cystic fibrosis mice in vivo following luminal application of DANN complexes targeted to the serpin-enzyme complex receptor. Mol Ther 2002; 5: 413–419.
Yonemitsu Y, Kitson C, Ferrari S, Farley R, Griesenbach U, Judd D et al. Efficient gene transfer to airway epithelium using recombinant Sendai virus. Nat Biotechnol 2000; 18: 970–973.
Griesenbach U, Cassady RL, Ferrari S, Fukumura M, Muller C, Schmitt E et al. The nasal epithelium as a factory for systemic protein delivery. Mol Ther 2002; 5: 98–103.
Sheppard DN, Welsh MJ . Structure and function of the CFTR chloride channel. Physiol Rev 1999; 79 (1 Suppl): S23–S45.
Wilson R, Alton E, Rutman A, Higgins P, Al Nakib W, Geddes DM et al. Upper respiratory tract viral infection and mucociliary clearance. Eur J Respir Dis 1987; 70: 272–279.
Liang L, He C, Lei M, Li S, Hao Y, Zhu H et al. Pathology of guinea pigs experimentally infected with a novel reovirus and coronavirus isolated from SARS patients. DNA Cell Biol 2005; 24: 485–490.
Chisari FV, Filippi P, Buras J, McLachlan A, Popper H, Pinkert CA et al. Structural and pathological effects of synthesis of hepatitis B virus large envelope polypeptide in transgenic mice. Proc Natl Acad Sci USA 1987; 84: 6909–6913.
Li HO, Zhu YF, Asakawa M, Kuma H, Hirata T, Ueda Y et al. A cytoplasmic RNA vector derived from nontransmissible Sendai virus with efficient gene transfer and expression. J Virol 2000; 74: 6564–6569.
Zhang L, Bukreyev A, Thompson CI, Watson B, Peeples ME, Collins PL et al. Infection of ciliated cells by human parainfluenza virus type 3 in an in vitro model of human airway epithelium. J Virol 2005; 79: 1113–1124.
Tabcharani JA, Linsdell P, Hanrahan JW . Halide permeation in wild type and mutant cystic fibrosis transmembrane conductance regulator chloride channels. J Gen Physiol 1997; 110: 341–354.
Gray MA, Harris A, Coleman L, Greenwell JR, Argent BE . Two types of chloride channel on duct cells cultured from human fetal pancreas. Am J Physiol 1989; 257: C240–C251.
Larsen EH, Price EM, Gabriel SE, Stutts MJ, Boucher RC . Clusters of Cl− channels in CFTR-expressing Sf 9 cells switch spontaneously between slow and fast gating modes. Pflugers Arch – Eur J Physiol 1996; 432: 528–537.
Mohammad-Panah R, Demolombe S, Riochet D, Leblais V, Loussouran G, Pollard H et al. Hyperexpression of recombinant CFTR in heterologous cells alters its physiological properties. Am J Physiol 1998; 274: C310–C318.
Hurwitz JL, Soike KF, Sangster MY, Portner A, Sealy RE, Dawson DH et al. Intranasal Sendai virus vaccine protects African green monkeys from infection with human parainfluenza virus-type I. Vaccine 1997; 15: 533–540.
Slobod KS, Shenep JL, Lujan-Zilbermann J, Allison K, Brown B, Scroggs RA et al. Safety and immunogenicity of intranasal murine parainfluenza virus type 1 (Sendai virus) in healthy human adults. Vaccine 2004; 22: 3182–3186.
Engelhardt JF, Yankaskas JR, Ernst SA, Yang Y, Marino CR, Boucher RA et al. Submucosal glands are the predominant site of CFTR expression in human bronchus. Nat Genet 1992; 2: 240–247.
Farmen SL, Karp PH, Ng P, Palmer DJ, Koehler DR, Hu J et al. Gene transfer of CFTR to airway epithelia: low levels of expression are sufficient to correct Cl− transport and overexpression can generate basolateral CFTR. Am J Physiol Lung Cell Mol Physiol 2005; 289: L1123–1130.
Schiavi SC, Abdelkader N, Reber S, Pennington S, Narayana R, McPherson JM et al. Biosynthetic and growth abnormalities are associated with high-level expression of CFTR in heterologous cells. Am J Physiol Cell Physiol 1996; 270: C341–C351.
Ye L, Chan S, Chow YH, Tsui LC, Hu J . Regulated expression of the human CFTR gene in epithelial cells. Mol Ther 2001; 3: 723–733.
Tokusumi T, Iida A, Hirata T, Kato A, Nagai Y, Hasegawa M . Recombinant Sendai virus expressing different levels of a foreign gene. Virus Res 2002; 86: 33–38.
Inoue M, Tokusumi Y, Ban H, Shirakura M, Kanava T, Yoshizaki M et al. Recombinant Sendai virus vectors deleted in both the matrix and the fusion genes: efficient gene transfer with preferable properties. J Gene Med 2004; 6: 1069–1081.
Yoshizaki M, Hironaka T, Iwasaki H, Ban H, Tokusumi Y, Iida A et al. Naked Sendai virus vector lacking all of the envelope-related genes: reduced cytopathogenicity and immunogenicity. J Gene Med 2006; 8: 1151–1159.
Griesenbach U, Mitomo K, Inoue M, Somerton L, Farley R, Singh C et al. Lentivirus pseudotyped with envelope proteins F and HN from Sendai virus transducer airway epithelium efficiently and persistently. Mol Ther 2006; 13 (Suppl 1) Abs 367: S139–S140.
Hasan MK, Kato A, Shioda T, Sakai Y, Yu D, Nagai Y . Creation of an infectious recombinant Sendai virus expressing the firefly luciferase gene from the 3′ proximal first locus. J Gen Virol 1997; 78: 2813–2820.
Sakai Y, Kiyotani K, Fukumura M, Asakawa M, Kato A, Shioda T et al. Accommodation of foreign genes into the Sendai virus genome: sizes of inserted genes and viral replication. FEBS Lett 1999; 456: 221–226.
Kato A, Sakai Y, Shioda T, Kondo T, Nakanishi M, Nagai Y . Initiation of Sendai virus multiplication from transfected cDNA or RNA with negative or positive sense. Genes Cells 1996; 1: 569–579.
Yonemitsu Y, Kaneda Y . Hemagglutinating virus of Japan-liposome-mediated gene delivery to vascular cells. In: Molecular Biology of Vascular Diseases. Method in Molecular Medicine. Human Press: Totowa, NJ, 1999, pp 295–306.
Denning GM, Anderson MP, Amara JF, Marshall J, Smith AE, Welsh MJ . Processing of mutant cystic fibrosis transmembrane conductance regulator is temperature-sensitive. Nature 1992; 358: 761–764.
Rotoli BM, Bussolati O, Dall’Asta V, Hoffmann EK, Cabrini G, Gazzola GC . CFTR expression in C127 cells is associated with enhanced cell shrinkage and ATP extrusion in Cl−-free medium. Biochem Biophys Res Commun 1996; 227: 755–761.
Wright AM, Gong X, Verdon B, Linsdell P, Mehta A, Riordan JR et al. Novel regulation of cystic fibrosis conductance regulator (CFTR) channel gating by external chloride. J Biol Chem 2004; 279: 41658–41663.
Venglarik CJ, Bridges RJ, Frizzell RA . A simple assay for agonist-regulated Cl and K conductances in salt-secreting epithelial cells. Am J Physiol 1990; 259: C358–C364.
Munkonge FM, Osborne LR, Geddes DM, Alton EWFW . Lack of inhibition by dideoxy-forskolin and verapamil of DIDS-sensitive volume-activated Cl− secretion in human squamous lung carcinoma epithelial cells. Biochim Biophys Acta 1994; 1224: 342–348.
Delaney SJ, Alton EWFW, Smith SN, Lunn DP, Farley R, Lovelock PK et al. Cystic fibrosis mice carrying the missense mutation G551D replicate human genotype-phenotype correlations. EMBO J 1996; 15: 955–963.
Zhou L, Dey CR, Wert SE, DuVall MD, Frizzell RA, Whitsett JA . Correction of lethal intestinal defect in a mouse model of cystic fibrosis by human CFTR. Science 1994; 266: 1705–1708.
Smith SN, Middleton PG, Chadwick S, Jaffe A, Bush KA, Rolleston S et al. The in vivo effects of Milrinone on the airways of cystic fibrosis mice and human subjects. Am J Respir Cell Mol Biol 1999; 20: 129–134.
Aherne WA, Dunnill MS . Point counting and morphometry. In: Aherne WA and Dunnill MS (eds). Morphometry. Arnold E: London, 1982, pp 33–45.
Acknowledgements
This study was supported by the UK Cystic Fibrosis Trust through a grant to the UK Cystic Fibrosis Gene Therapy Consortium (www.cfgenetherapy.org.uk) and a CF Trust Senior Benjamin Angel Fellowship (UG).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ferrari, S., Griesenbach, U., Iida, A. et al. Sendai virus-mediated CFTR gene transfer to the airway epithelium. Gene Ther 14, 1371–1379 (2007). https://doi.org/10.1038/sj.gt.3302991
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302991
Keywords
This article is cited by
-
Viral and Synthetic RNA Vector Technologies and Applications
Molecular Therapy (2016)
-
Validation of recombinant Sendai virus in a non-natural host model
Gene Therapy (2011)
-
Pushing the envelope in the lung
Gene Therapy (2011)
-
Gene therapy: light is finally in the tunnel
Protein & Cell (2011)
-
Toward Gene Therapy for Cystic Fibrosis Using a Lentivirus Pseudotyped With Sendai Virus Envelopes
Molecular Therapy (2010)