Abstract
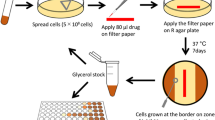
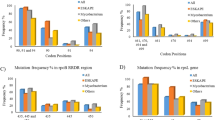
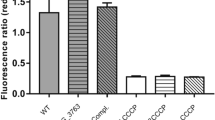
Ethambutol (EMB), a frontline antituberculous drug, targets the mycobacterial cell wall, a unique structure among prokaryotes which consists of an outer layer of mycolic acids covalently bound to peptidoglycan via the arabinogalactan. EMB inhibits the polymerization of cell wall arabinan, and results in the accumulation of the lipid carrier decaprenol phosphoarabinose1,2, which suggests that the drug interferes with the transfer of arabinose to the cell wall acceptor. Unfortunately, resistance to EMB has been described in up to 4% of clinical isolates of Mycobacterium tuberculosis and is prevalent among isolates from patients with multidrug-resistant tuberculosis3. We used resistance to EMB as a tool to identify genes participating in the biosynthesis of the mycobacterial cell wall. This approach led to the identification of the embCAB gene cluster, recently proposed to encode for mycobacterial arabinosyl transferases4. Resistance to EMB results from an accumulation of genetic events determining overexpression of the Emb protein(s), structural mutation in EmbB, or both. Further characterization of these proteins might provide information on targets for new chemotherapeutic agents and might help development of diagnostic strategies for the detection of resistant M. tuberculosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Takayama, K. & Kilburn, J.O. Inhibition of synthesis of arabinogalactan by ethambutol in Mycobacterium smegmatis. Antimicrob. Agents Chemother. 33, 1493–1499 (1989).
Wolucka, B.A., McNeil, M.R., de Hoffmann, E., Chojnacki, T. & Brennan, P.J. Recognition of the lipid intermediate for arabinogalactan/arabinomannan biosynthesis and its relation to the mode of action of ethambutol on mycobacteria. J. Biol. Chem. 269, 23328–23335 (1994).
Bloch, A.B. et al. Nationwide survey of drug-resistant tuberculosis in the United States. JAMA 271, 665–671 (1994).
Belanger, A.E. et al. The embAB genes of Mycobacterium avium encode an arabinosyl transferase involved in cell wall arabinan biosynthesis that is the target for the an-timycobacterial drug ethambutol. Proc. Natl. Acad. Sd. USA 93, 11919–11924 (1996).
Snapper, S.B., Melton, R.E., Mustafa, S., Kieser, T. & Jacobs, W.R. Jr. Isolation and characterization of efficient transformation mutants of Mycobacterium smegmatis. Mol. Microbiol. 4, 1911–1919 (1990).
Eiglmeier, K., Honoré, N., Woods, S.A., Caudron, B. & Cole, S.T. Use of an ordered library to deduce the genomic organization of Mycobacterium leprae. Mol. Microbiol. 7, 197–206 (1993).
Maddry, J.A., Suling, W.J. & Reynolds, R.C. Glycosyl transferases as targets for inhibition of cell wall synthesis in M tuberculosis and M. avium. Res. Microbiol. 147, 106–112 (1996).
Deng, L. et al. Recognition of multiple effects of ethambutol on metabolism of mycobacterial cell envelope. Antimicrob. Agents Chemother. 39, 694–701 (1995).
Musser, J.M. Antimicrobial agent resistance in mycobacteria: Molecular genetic insights . Clin. Microbiol. Rev. 8, 496–514 (1995).
Telenti, A. Genetics of drug resistance in tuberculosis. Clin. Chest Med. 18, 5.1–5.10 (1997).
Zhang, Y., Heym, B., Allen, B., Young, D. & Cole, S.T. The catalase-peroxidase gene and isoniazid resistance of Mycobacterium tuberculosis. Nature 358, 501–593 (1992).
Banerjee, A. et al. inhA, a gene encoding a target for isoniazid and ethionamide in Mycobacterium tuberculosis. Science 263, 227–230 (1994).
Deretic, V. et al. Mycobacterium tuberculosis is a natural mutant with an inactivated oxidative-stress regulatory gene: Implications for sensitivity to isoniazid. Mol. Microbiol. 17, 889–900 (1995).
Wilson, T.M. & Collins, D.M. ahpC, a gene involved in isoniazid resistance of the Mycobacterium tuberculosis complex. Mol. Microbiol. 19, 1025–1034 (1996).
Telenti, A. et al. Genotypic assessment of isoniazid and rifampin resistance in Mycobacterium tuberculosis: A blind study at reference laboratory level. J. Clin. Microbiol. 35, 719–723 (1997).
Telenti, A. et al. Detection of rifampicin-resistance mutations in Mycobacterium tuberculosis. Lancet 341, 647–650 (1993).
Finken, M., Kirschner, P., Meier, A., Wrede, A. & Bottger, E.C. Molecular basis of streptomycin resistance in Mycobacterium tuberculosis: Alterations of the ribosomal protein S12 gene and point mutations within a functional 16S ribosomal RNA pseudoknot. Mol. Microbiol. 9, 1239–1246 (1993).
Honoré, N. & Cole, S.T. Streptomycin resistance in mycobacteria. Antimicrob. Agents Chemother. 38, 238–242 (1994).
Takiff, H.E. et al. Cloning and nucleotide sequence of Mycobacterium tuberculosis gyrA and gyrB genes and detection of quinolone resistance mutations. Antimicrob. Agents Chemother. 38, 773–780 (1994).
Kocagoz, T. et al. Gyrase mutations in laboratory selected, fluoroquinolone-resistant mutants of Mycobacterium tuberculosis H37Ra. Antimicrob. Agents Chemother. 40, 1768–1774 (1996).
Takiff, H.E. et al. Efflux pump of the proton antiporter family confers low-level fluoroquinolone resistance in Mycobacterium smegmatis. Proc. Natl. Acad. Sci. USA 93, 362–366 (1996).
Scorpio, A. & Zhang, Y. Mutations in pncA, a gene encoding pyrazinamidase/nicotinamidase, cause resistance to the antituberculous drug pyrazinamide in tubercle bacillus. Nature Med. 2, 662–667 (1996).
Telenti, A. & Persing, D.H. Novel strategies for the detection of drug resistance in Mycobacterium tuberculosis. Res. Microbiol. 147, 73–79 (1996).
Lazlo, A., Rahman, M., Raviglione, M., Bustreo, F. & the WHO/IUTLD network of supranational reference laboratories. Quality assurance programme for drug susceptibility testing of Mycobacterium tuberculosis in the WHO/IUTLD supranational laboratory network: First round of proficiency testing. Intl. J. Tuberculosis Lung Dis. (in the press).
Gangadharam, P.R. & Gonzalez, E.R. Influence of the medium on the in vitro susceptibility of Mycobacterium tuberculosis to ethambutol. Am. Rev. Resp. Dis. 102, 653–655 (1970).
Heifets, L.B., Iseman, M.D. & Lindholm-Levy, P.J. Ethambutol MICs and MBCs for Mycobacterium avium complex and Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 30, 927–932 (1986).
Levin, M.E. & Hatfull, G.F. Mycobacterium smegmatis RNA polymerase: DNA super-coiling, action of rifampicin and mechanism of rifampicin resistance. Mol. Microbiol. 8, 277–285 (1993).
Rost, B. & Sander, C. Combining evolutionary information and neural networks to predict protein secondary structure. Proteins 19, 55–72 (1994).
Philipp, W.J. et al. An integrated map of the genome of the tubercle bacillus, Mycobacterium tuberculosis H37Rv, and comparison with M. leprae. Proc. Natl. Acad. Sci. USA 93, 3132–3137 (1996).
Telenti, A., Imboden, P., Marchesi, F., Schmidheini, T. & Bodmer, T., Direct, automated detection of rifampin-resistant Mycobacterium tuberculosis by polymerase chain reaction and single-strand conformation polymorphism. Antimicrob. Agents Chemother. 37, 2054–2058 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Telenti, A., Philipp, W., Sreevatsan, S. et al. The emb operon, a gene cluster of Mycobacterium tuberculosis involved in resistance to ethambutol. Nat Med 3, 567–570 (1997). https://doi.org/10.1038/nm0597-567
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0597-567
This article is cited by
-
Development of DNA Bio-chip for Detection of Mutations of rpoB, embB and inhA Genes in Drug-Resistant Mycobacterium Tuberculosis
Indian Journal of Clinical Biochemistry (2023)
-
Tuberculosis: current scenario, drug targets, and future prospects
Medicinal Chemistry Research (2021)
-
Application of Mycobacterium smegmatis as a surrogate to evaluate drug leads against Mycobacterium tuberculosis
The Journal of Antibiotics (2020)
-
Modeling the mechanism pathways of first line drug in Tuberculosis using Petri nets
International Journal of System Assurance Engineering and Management (2020)
-
Application of next-generation sequencing to detect variants of drug-resistant Mycobacterium tuberculosis: genotype–phenotype correlation
Annals of Clinical Microbiology and Antimicrobials (2019)