Article Text
Abstract
Rationale While cross-sectional studies have shown associations between certain occupational exposures and lower levels of lung function, there was little evidence from population-based studies with repeated lung function measurements.
Objectives We aimed to investigate the associations between occupational exposures and longitudinal lung function decline in the population-based Tasmanian Longitudinal Health Study.
Methods Lung function decline between ages 45 years and 50 years was assessed using data from 767 participants. Using lifetime work history calendars completed at age 45 years, exposures were assigned according to the ALOHA plus Job Exposure Matrix. Occupational exposures were defined as ever exposed and cumulative exposure -unit- years. We investigated effect modification by sex, smoking and asthma status.
Results Compared with those without exposure, ever exposures to aromatic solvents and metals were associated with a greater decline in FEV1 (aromatic solvents 15.5 mL/year (95% CI −24.8 to 6.3); metals 11.3 mL/year (95% CI −21.9 to – 0.7)) and FVC (aromatic solvents 14.1 mL/year 95% CI −28.8 to – 0.7; metals 17.5 mL/year (95% CI –34.3 to – 0.8)). Cumulative exposure (unit years) to aromatic solvents was also associated with greater decline in FEV1 and FVC. Women had lower cumulative exposure years to aromatic solvents than men (mean (SD) 9.6 (15.5) vs 16.6 (14.6)), but greater lung function decline than men. We also found association between ever exposures to gases/fumes or mineral dust and greater decline in lung function.
Conclusions Exposures to aromatic solvents and metals were associated with greater lung function decline. The effect of aromatic solvents was strongest in women. Preventive strategies should be implemented to reduce these exposures in the workplace.
- solvents
- chronic obstructive pulmonary disease
- job exposure matrix
- occupational exposure
- lung function
- FEV1
Statistics from Altmetric.com
- solvents
- chronic obstructive pulmonary disease
- job exposure matrix
- occupational exposure
- lung function
- FEV1
Key messages
What is the key question?
What are the associations between occupational exposures and lung function decline in the general population?
What is the bottom line?
This population-based longitudinal study showed associations between different classes of occupational exposures and greater lung function decline between the ages of 45 years and 50 years. In particular, both ever and cumulative exposures to aromatic solvents were associated with greater lung function decline. Lung function decline associated with aromatic solvents was higher in women compared with men, even though they had been less exposed.
Why read on?
Our findings on the aromatic solvents are novel and highlight the need for strengthening preventive strategies aimed at reducing exposure to aromatic solvents in the workplace.
INTRODUCTION
Adult lung function decline is a normal feature of ageing, but excessive decline during adulthood can result in fixed airflow obstruction and is a predictor of higher mortality and morbidity.1 Identification of modifiable risk factors for early or excessive lung function decline, therefore has important public health implications. However, the evidence of such factors is scarce due to a limited number of general population studies with repeated tracking of lung function measurements over time.
Occupational exposures, mainly vapours, gases, dust and fumes (VGDF)2–7 and pesticides, have been investigated as risk factors for lung function decline.2 While some have observed these exposures to have detrimental effects on lung function decline, others have contradicted these findings.8 9 Two other common occupational exposures gaining increasing recognition as having an impact on lung function are solvents and metals.10–12 Recently, in a cross-sectional analysis, we identified an association between occupational exposures to solvents and metals with fixed airflow obstruction and lower lung function.13 Other cross-sectional studies have also suggested this relationship, but none of the published studies to date have been able to establish temporal relationships between these exposures and airflow obstruction and lung function.11 14–16
Previous population-based studies that assessed self-reported occupational exposures and lung function decline have been limited by the use of current or most recent job to define occupational exposure, which is subject to information and selection bias.4 5 7 Job Exposure Matrices (JEMs) provide some protection from these types of bias, because they are applied consistently to reported job titles and do not rely on self-assessed exposures. Furthermore, JEM applied to complete work history calendars can assess lifetime exposures capturing multiple jobs and job transitions over the life course.17 However, few population-based studies have used JEM to investigate the associations between occupational exposures and lung function decline over time, and none have investigated exposures to solvents and decline of lung function.2 8 18
Given the discrepancies between and limitations of the studies conducted to date, we have investigated the association between occupational exposure and lung function decline using a general population-based cohort. We assessed occupational exposures from lifetime work history calendars to identify and calculate ever exposure and cumulative exposure using a general population-based JEM. We also investigated whether sex, asthma or smoking status modified these relationships.
Methods
Study design
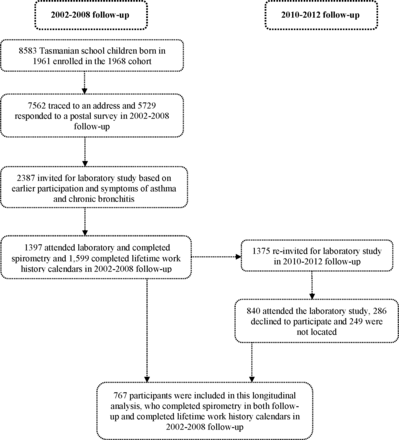
Our study included participants from 2002 –2008 and 2010 –2012 follow-up surveys of the Tasmanian Longitudinal Health Study (TAHS) for which methodology and previous results have been published elsewhere.19 Briefly, TAHS began in 1968 by recruiting all 8583 school children born in 1961 in Tasmania. In 2002, we traced 7562 (88.1%) participants to a residential address, and 5729 (78.4%) responded to a postal survey. A subsample (n=2387) of these participants in 2004, selected by participation in previous follow-ups and enriched for participants with a history of asthma and bronchitis, were invited to take part in a clinical study and 1397 (58.6%) participated.
In 2010, a new follow-up was conducted on the laboratory attendees from the 2004 clinical study participants. Of 1397 who were eligible, 840 (61.1%) took part in a full laboratory visit. (figure 1).19 In the current analysis, we included 767 participants who completed lung function at both follow-ups and a lifetime work history calendar during the 2002–2008 follow-up study. All participants provided written informed consent.
Flow chart illustrating study design.
Lung function measurements
The rate of lung function decline was calculated as FEV1 (mL/year) (∆FEV1), FVC (∆FVC) and FEV1/FVC (∆FEV1/FVC) using the change in each measure divided by the time interval (age in years) between follow-ups for participants who completed lung function testing in both follow-ups and a work history calendar during the 2002–2008 follow-up. Both prebronchodilator (pre-BD) and post-BD spirometry were collected in first follow-up (2002–2008), but in the 2010–2012 follow-up we did not have the post-BD measurement. Therefore, this analyses was only able to investigate using the pre-BD lung function. Pre-BD spirometry at both time points was performed using the EasyOne Pro Ultrasonic Spirometer (ndd, Medizintechnik, AG, Switzerland) using the same methods. Spirometry was conducted according to the American Thoracic Society and European Respiratory Society joint statement.20 Detailed methods have been reported elsewhere.21
Occupational exposures
Occupational exposure was classified using the lifetime work history calendars that were collected from the participants from the 2002–2008 follow-up. Participants were requested to list all jobs held in their lifetime including job title, industry description, employer description, year work started and ended. The job titles were standardised according to the International Standard Classification of Occupations-88 four-digit classification.22 These classifications were linked to a general population-based JEM called the ALOHA plus JEM23 and assigned exposures into biological dust, mineral dust, gases/fumes, VGDF, fungicides, herbicides, insecticides, aromatic solvents, chlorinated solvents, other solvents and metals. In case participants had two different jobs at the same time, exposures of both jobs were averaged and allocated half of the exposure. We assigned people as having ever exposure if they had exposure for any job over their working life. We combined low-exposure and high-exposure categories to define as ever exposure because of the small number of participants in the high-exposure group limiting the statistical analysis. We also calculated cumulative exposure-unit (EU) years by multiplying the number of years worked in a given job and exposure intensity (weighted by 4 for high exposure and 1 for low exposure) for the given job.24 This was then summed for each individual and each exposure.
Definitions of key confounders
Having had asthma was defined by an affirmative response to the question ‘Have you ever had asthma?’. Current asthma was based on self-reported symptoms in the morning, daytime, and night-time or flare-ups in the last 12 months. Childhood asthma was defined as asthma reported at age 7 years and 13 years by an affirmative response by the parents to the question ‘Has he/she at any time of his/her life suffered from attacks of asthma or wheezy breathing?’. Smoking status of the participants was categorised as current smokers, past smokers and never smokers. A current smoker was defined as smoking status within the last 4 weeks. Pack-years were calculated as the number of cigarettes smoked per day divided by 20 multiplied by the number of years of smoking.24 Childhood and adulthood socioeconomic statuses were defined using Australian census data of Socio-Economic Indexes for Areas.25 We used the Index of Education and Occupation (IEO) variables that focused on the skills of the people required in different occupations. A high IEO score indicated that people who lived in that area had high qualifications and highly skilled jobs.
Statistical analysis
We used Stata V.15.0 for Windows (Stata Corporation, College Station, Texas, USA) for all statistical analyses. Seven hundred and sixty-seven participants with complete lung function test and work history calendar were included in this analysis using multiple linear regression (detail in the online supplementary material). The estimates were calculated using inverse-probability-of-inclusion weights to adjust for the enriched sample of the 2002 –2008 follow-up. We adjusted for sex, height, smoking status, pack-years (from both follow-ups), childhood asthma, current asthma and socioeconomic status in both childhood and adulthood.
Supplemental material
We additionally adjusted the models with biological dust, mineral dust and gases/fumes for all types of pesticides, the models with fungicides, herbicides and insecticides were additionally adjusted for combined exposure to VGDF, while the models with solvents and metals were additionally adjusted for all types of pesticides and VGDF due to the presence of correlation (online supplementary figure E1 and online supplementary figure E2). Furthermore, three categories of solvents (aromatic, chlorinated and other solvents) were included one by one as an additional confounder in the adjusted regression model with each solvent exposure (for example; chlorinated and other solvents were added to the model with aromatic solvents) to observe the change in the effect estimates. As this model does not alter the key message of this paper, we did not adjust for these additional confounders in the main analysis.
Supplemental material
Supplemental material
We also tested for effect modification by sex, smoking status and asthma on the associations between occupational exposure and lung function decline. We compared the models with or without effect modification, and the p value was reported using the likelihood ratio test. We did not find any effect modification by smoking status and asthma on the association between cumulative EU-years and lung function decline, but we did observe effect modification by sex.
We also performed multiple imputation to deal with missing data. Results from imputation models were compared with those derived from complete case analysis (see online supplementary material for more details). We have conducted several sensitivity analyses such as adjusting for baseline pre-BD lung function and using random-effects linear regression models. We have also presented our cumulative exposure results without weighting by exposure intensity (see online supplementary material for more details). A value of p<0.05 was considered as statistically significant for all models including interaction analysis.
Results
Population characteristics
The characteristics of study participants, smoking history and lung function decline are given in table 1. Half of the 767 participants who took part in both clinical follow-ups were men (50.0%). The average follow-up time was 5 years. Between the follow-up visits, active smoking decreased from 20% to 15% (table 1). The mean lung function decline in the FEV1/FVC ratio was 0.6%, indicating that FEV1 declined more rapidly than FVC.
Study participant characteristics and lung function (n=767)
The prevalences of occupational exposures are presented in online supplementary table E1. The most common exposures were gases/fumes (70%), biological dust (53%), mineral dust (50%), aromatic solvents (29%) and metals (22%).
Ever exposure to occupational exposures and lung function decline
Table 2 shows the univariable and multivariable association between ever exposure and lung function decline. Those who were ever exposed to gases/fumes (mean=31.2, SE=2.04, (online supplementary table E2) had a significantly greater decline in FEV1 (11.4 mL/year, 95% CI −20.0 to to 2.9) compared with those not exposed to gases/fumes (mean=19.5, SE=3.7, (online supplementary table E2)).
Univariable and multivariable associations between ever exposures and lung function decline (N=767)
Ever exposure to aromatic solvents was associated with a greater decline in FEV1 (15.5 mL/year, 95% CI −24.8 to -6.3) and FVC (14.1 mL/year, 95% CI −28.8 to -0.7), but not FEV1/FVC ratio. Ever exposure to metals was associated with a greater decline in FEV1 (11.3 mL/year, 95% CI −21.9 to -0.7) and FVC (17.5 mL/year, 95% CI −34.3 to -0.8), but not the FEV1/FVC ratio. Ever exposure to chlorinated solvents was associated with greater decline in FEV1 (11.6 mL/year, 95% CI −21.5 to 1.6) but not the FVC and FEV1/FVC ratio. Further adjustments for other two categories of solvents (online supplementary table E3) and baseline pre-BD lung function (online supplementary table E4) did not change these findings. Ever exposure to mineral dust (0.2%/year, 95% CI −0.3 to -0.02) and gases/fumes (0.2%/year, 95% CI −0.4 to -0.1) were associated with a greater decline in FEV1/FVC ratio.
No significant associations were found for fungicides, herbicides and insecticides with any of the outcomes (table 2 and online supplementary table E5).
Cumulative exposures and lung function decline
We have presented median (Q1, Q3) cumulative EU-years in online supplementary table E1. Median cumulative EU-years was highest (19.5 (7.5–52) years) for chlorinated solvents, metals (19 (6–48.5)years), gases/fumes (19 (8–33) years) followed by biological dust, mineral dust, insecticides, other solvents and aromatic solvents.
The multivariable associations between cumulative EU-years and lung function decline are presented in table 3. Per EU-year increase in cumulative exposure to aromatic solvents, there was a 0.6 mL/year (95% CI −1.0 to 0.3) greater decline in FEV1 and a 0.9 mL/year (95% CI −1.6 to 0.4) greater decline in FVC. We also found a significant association between cumulative EU-years to gases/fumes and decline in FEV1. These findings were slightly attenuated, but consistent when unweighted cumulative exposures were added to the models (online supplementary table E6). We also observed associations with cumulative EU-years to fungicides and insecticides and higher FVC but did not observe any associations between cumulative EU-years to any other exposures and lung function (table 3 and online supplementary table E7).
Multivariate association between cumulative exposure-unit (EU)- years and lung function decline (N=723)
Effect modification
We examined whether the relationships between occupational exposures and lung function decline were modified by sex, asthma and smoking status. Neither asthma nor smoking status modified any association (online supplementary table E8). However, we observed the association between cumulative EU-years to aromatic solvents and greater decline in FEV1 to be stronger in women than men (p for interaction=0.02) and the FEV1/FVC ratio (p for interaction=0.01) (table 4). On the other hand, women had less cumulative exposure to aromatic solvents (mean±SD = 9.6±15.5) compared with men (mean±SD = 16.6±14.6). Figures 2–4 display the predicted declines in FEV1, FVC and FEV1/FVC ratio that were calculated using linear estimates from multiple linear regression models. In this model, women with increasing cumulative EU-years to aromatic solvents had a greater lung function decline. However, we did not observe any such relationship for men. We did not observe sex to modify the associations related to any of the other exposures.
The association between cumulative years of exposure to aromatic solvents and linear predicted decline in FEV1, stratified by sex. Circles represent the decline of the FEV1 and the bars represent the 95% CI.
The association between cumulative years of exposure to aromatic solvents and linear predicted decline in FVC, stratified by sex. Circles represent the decline of the FVC and the bars represent the 95% CI.
The association between cumulative years of exposure to aromatic solvents and linear predicted decline in FEV1/FVC ratio, stratified by sex. Circles represent the decline of the FEV1/FVC ratio and the bars represent the 95% CI.
Effect modification by sex on the relationship between cumulative EU-years to solvents and lung function declines (N=723)
Discussion
In this longitudinal study, we have shown multiple occupational exposures to be associated with greater lung function decline between the ages of 45 years and 50 years. Importantly, we identified greater lung function decline, with ever being exposed and cumulative EU-years to aromatic solvents. In particular, we observed that women exposed to aromatic solvents had a greater decline in both FEV1 and FEV1/FVC ratio compared with men. We also observed a greater decline in lung function for those ever exposed to gases/ fumes, chlorinated solvents or metals, but not for mineral dust, biological dust, fungicides, herbicides or insecticides.
This longitudinal study provides robust evidence that both ever exposures and cumulative exposures to aromatic solvents are significantly associated with greater lung function decline in a general population sample. Cross-sectional studies have provided some evidence for an association between solvents and lower levels of lung function,14 23 but the findings were contradictory. One cross-sectional study from France found an association between specific solvents (benzene, carbon tetrachloride, degreasing agents, other solvents and epoxy resins) and lower lung function.14 Another cross-sectional study from the Netherlands did not find any association between exposure to solvents and lung function.23 Several cross-sectional studies11 26–28 and one longitudinal study10 suggested that solvent exposure was associated with self-reported respiratory symptoms and chronic bronchitis. Although a study of the Zutphen cohort used lifetime work histories to assign exposure to solvents and showed that it was related to chronic non-specific lung disease, no information was reported on lung function decline.10 In our analysis we controlled for a range of confounders including coexposures and found that exposure to solvents is an important risk factor for lung function decline.
Cumulative EU-years to aromatic solvents was associated with greater decline in FEV1 and FVC. However, we did not find the same effects for cumulative EU-years to chlorinated or other solvents. There may be some reasons for this difference between the categories of solvents. It could be related to nature and chemical structure of the solvents. For example, some aromatic solvents (eg, benzene) are absorbed better than chlorinated solvents.29 The differences in the job distribution between aromatic and chlorinated solvents may be another explanation for the variation across solvents. For example, a large proportion of our sample (both men and women) exposed to aromatic but not chlorinated solvents were working in painting, electrical, mining and construction industries, while participants exposed to chlorinated solvents were working as hand-launderers, pressers, metal, machinery and related trade industries.30 Exposures to solvents also depend on the vapour concentration in the workplace and actual cumulative duration worked by the participants. The complex interplay between these factors may also influence lung function decline to different types of solvents.29
Our study was the first to investigate different effects of solvent exposure on lung function in men and women. We found that despite women having lower cumulative EU-years to aromatic solvents compared with men, among women increasing cumulative EU-years to aromatic solvents was associated with greater decline in FEV1 and FEV1/FVC ratio. However, this was not observed among men, despite women having lower cumulative EU-years to aromatic solvent compared with men. This sex difference may be related to the differences in the patterns of occupational exposures.31 32 Women in our study exposed to aromatic solvents were sculptors/painters/artists, woodworkers and life science technicians, while men were mostly painters, carpenters, plumbers, pipe fitters and firefighters.
Furthermore, potential influences of female sex hormones may also explain the above sex difference.33 Toxins in the airways are metabolised via the cytochrome P450 pathway.31 The female sex hormone oestrogen can influence this pathway by causing the upregulation of the CYP1A1, CYP1A2, CYP2A6, CYP3A4 and CYP1B1 cytochromes. The role of these enzymes in the toxin metabolism pathway leads to increased oxidation of inhaled substances leading to increased oxidative stress in the airways and greater risk of airflow obstruction in women.34 A few cross-sectional studies conducted on different occupational exposures reported a significant association with airflow obstruction as measured by lung function in women but not men.24 35 To our knowledge, previous population-based longitudinal studies that examined the potential effect modification of the association between occupational exposures and lung function decline by sex did not find any difference between men and women.2 8 In our study, we also did not find any difference between men and women ever exposed to solvents, but we found an association for women with increasing cumulative EU-years.
Our analysis shows an association between ever exposure and cumulative EU-years to gases/fumes and a greater decline in FEV1. The European Community Respiratory Health Survey8 and the Vlagtwedde-Vlaardingen Cohort Study2 used the ALOHA JEM to assess occupational exposure, but the authors did not find an association between ever exposure (low or high) to gases/fumes with lung function decline. While the Vlagtwedde-Vlaardingen Study found a borderline significant association with cumulative EU-years to combined exposure to VGDF, this association disappeared after adjustment for possible coexposure to pesticides.2 The Vlagtwedde-Vlaardingen Study also used the current job to assign exposure, and there is a possibility that this study underestimated the true impact of gases/fumes exposure on lung function decline. Our study used lifetime work history calendars across an individual’s life span, and our association remained even after adjustment for coexposure to all type of pesticides.
We also found an association between ever exposure to metals and greater decline in FEV1 and FVC. These findings are consistent with a workplace-based longitudinal study that found an association between cobalt dust exposure and lung function decline.36 Subjects exposed to metals in our study described themselves as working in metal or steel industries such as motor vehicle mechanics and fitters, sheet metal workers, welders and flame cutters and agriculture or industrial machinery mechanics. A recent meta-analysis which included longitudinal studies of welders also found an association between exposure to welding fumes and lung function decline.37 This raises the possibilities that the observed association with ever exposure to metal could be related to exposures in the specific industries such as smelting furnace fumes. However, there is also a chance of inhalation of metal in the form of dust through fettling and grinding processes that may cause lung function decline.
Our study had several strengths including an extensive data set that allowed adjusting for a wide range of confounders including pack-years of smoking, childhood and adult asthma, baseline lung function, socioeconomic status at the age of 7 years, 12 years and 45 years, and coexposures. In contrast, no previous longitudinal study has been able to adjust for all the identified confounding variables because of lack of early life exposure data.2 8 Adjustment of coexposures further strengthened this study by minimising the chance of overlapping exposures between three categories of solvents. We also used two estimates of occupational exposure: lifetime exposure and cumulative EU-years. Our exposure was classified based on the ALOHA plus JEM, which is specifically designed for general population-based studies. An advantage of using JEM-based estimates is that they are less likely to be affected by recall bias or differential misclassification.17
Despite these strengths, limitations of our study include the possibility of non-differential misclassification due to a potential heterogeneity of JEM-based exposure within a given job title. Such misclassification typically biases the estimates towards the null, resulting in an underestimation of the true effect. Occupational histories were collected at the 2002–2008 follow-up, and there is a chance that participants with respiratory or lung function problems could have changed their occupations during the follow-up time from a highly exposed to less exposed job (or vice versa) or changed their work environment to reduce the level of exposure. This may result in underestimation of the observed associations between occupational exposure and lung function. Although we have adjusted our analysis for both smoking and pack-years, the chance of residual confounding by smoking remains a possibility.
Another limitation is that the average follow-up period for this analysis was 6 years, which is lower than other cohort studies.2 8 Additionally, the smaller number of participants with work history calendar and spirometry in our study also reduced the sample size in this analysis and therefore the power to detect small effects. Also, by design, participants at baseline (2002–2008 follow-up) were enriched for asthma and bronchitis, which might affect the generalisability of our results. However, this enrichment is unlikely to affect the presented associations, as the addition of sampling weights to our analytical models did not significantly change these findings. The associations we have observed with lung function decline are biologically plausible13 17 21 and are consistent with previous epidemiological evidence.2 3 6 However, based on the limitations we suggest that our novel findings on the association between aromatic solvents and lung function decline should not be considered yet as causal until replication in a larger cohort is undertaken.
In conclusion, we have shown for the first time that exposures to aromatic solvents are associated with greater decline in lung function and confirmed the previous findings on the impact of metals, gases/fumes and mineral dust on lung function decline. Our findings provide novel insights into effect modification by sex on the relationship between cumulative exposures to aromatic solvents and greater lung function decline. Our findings suggest that the effect of aromatic solvents is not due to greater exposure in women, but due to greater susceptibility. This greater lung function decline in women may subsequently increase the risk of obstructive airway disease in future.
Acknowledgments
The authors thank the Tasmanian Longitudinal Health Study (TAHS) participants and previous investigators. The authors also thank Professor Mark Jenkins, PhD, Centre Director, Centre for Epidemiology and Biostatistics, School of Population and Global Health, The University of Melbourne, Victoria, who is a TAHS investigator but not a coauthor of this manuscript, for his assistance with obtaining funds and data collection. The authors also thank all the respiratory scientists who collected data in the lung function laboratories of Tasmania, Victoria, Queensland, and New South Wales; the research interviewers and data entry operators; and Ms Cathryn Wharton and Dr Desiree Mészáros for their organisational roles. The authors also thank the Archives Office of Tasmania for providing data from the 1968 TAHS questionnaires.
References
Footnotes
SD and MCM are joint senior authors.
Contributors Study concept and design: SMA, SD, GB, RV, HK, EHW, MJA, MCM. Acquisition of data: SMA, SD, PST, RW-B, BT, IF, RV, HK, EHW, MJA, MCM. Analysis and interpretation of data: SMA, SD, GB, JLP, MD, RV, HK, MJA, MCM. Drafting of the manuscript: SMA, SD, GB, MD, MCM. Critical revision of the manuscript for important intellectual content: all authors. Statistical analysis: SMA, MCM. Obtained funding: SD, GB, DPJ, LG, PST, JLH, RW-B, BT, IF, EHW, MJA, MCM. Study supervision: SM, SD, GB, MD, MCM. SMA and MCM had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Funding This study was supported by the National Health and Medical Research Council (NHMRC) of Australia under the NHMRC project grant scheme (299901, 1021275) and NHMRC European collaborative grant scheme (1101313) as part of ALEC (Ageing Lungs in European Cohorts) funded by the European Union’s Horizon 2020 research and innovation programme under grant agreement No 633212; The University of Melbourne; Clifford Craig Medical Research Trust of Tasmania; the Victorian, Queensland and Tasmanian Asthma Foundations; The Royal Hobart Hospital; Helen MacPherson Smith Trust; and GlaxoSmithKline. The funding agencies had no direct role in the conduct of the study, the collection, management, statistical analysis and interpretation of the data, preparation or approval of the manuscript. ISGlobal is a member of the CERCA Programme, Generalitat de Catalunya.
Competing interests None declared.
Patient consent for publication Obtained.
Ethics approval Both follow-ups were approved by the Human Research Ethics Committee of the University of Melbourne (HREC Ref. numbers 040375.1 and 0932983.1, respectively).
Provenance and peer review Not commissioned; externally peer reviewed.
Data sharing statement All data relevant to the study are included in the article or uploaded as supplementary information.
Linked Articles
- Airwaves
- Editorial