Article Text
Abstract
Background Although diabetes mellitus is implicated in susceptibility to infection, the association of diabetes with the subsequent course and outcome is unclear.
Methods A retrospective analysis of two multicentre cohorts was carried out. The effect of pre-existing diabetes on the host immune response, acute organ function and mortality in patients hospitalised with community-acquired pneumonia (CAP) in the GenIMS study (n=1895) and on mortality following either CAP or non-infectious hospitalisations in the population-based cohort study, Health ABC (n=1639) was determined. Measurements included the mortality rate within the first year, risk of organ dysfunction, and immune responses, including circulating inflammatory (tumour necrosis factor, interleukin 6, interleukin 10), coagulation (Factor IX, thrombin–antithrombin complexes, antithrombin), fibrinolysis (plasminogen-activator inhibitor-1 and D-dimer) and cell surface markers (CD120a, CD120b, human leucocyte antigen (HLA)-DR, Toll-like receptor-2 and Toll-like receptor-4).
Results In GenIMS, diabetes increased the mortality rate within the first year after CAP (unadjusted HR 1.41, 95% CI 1.12 to 1.76, p=0.002), even after adjusting for pre-existing cardiovascular and renal disease (adjusted HR 1.3, 95% CI 1.03 to 1.65, p=0.02). In Health ABC, diabetes increased the mortality rate within the first year following CAP hospitalisation, but not after hospitalisation for non-infectious illnesses (significant interaction for diabetes and reason for hospitalisation (p=0.04); HR for diabetes on mortality over the first year after CAP 1.87, 95% CI 0.76 to 4.6, p=0.16, and after non-infectious hospitalisation 1.16, 95% CI 0.8 to 1.6, p=0.37). In GenIMS, immediate immune response was similar, as evidenced by similar circulating immune marker levels, in the emergency department and during the first week. Those with diabetes had a higher risk of acute kidney injury during hospitalisation (39.3% vs 31.7%, p=0.005) and they were more likely to die due to cardiovascular and kidney disease (34.4% vs 26.8% and 10.4% vs 4.5%, p=0.03).
Conclusions Pre-existing diabetes was associated with a higher risk of death following CAP. The mechanism is not due to an altered immune response, at least as measured by a broad panel of circulating and cell surface markers, but may be due to worsening of pre-existing cardiovascular and kidney disease.
- Pneumonia
Statistics from Altmetric.com
It is a long-standing medical axiom that diabetes mellitus is a risk factor for infection.1 2 However, once infection occurs, the effect of diabetes on mortality is less clear. Some studies suggest that diabetes is associated with higher mortality after an infection, but others show no association.3–7 Previous studies that showed the association between diabetes and higher mortality may be confounded by higher prevalence of pre-existing chronic conditions, such as chronic kidney disease (CKD) or cardiovascular disease.8–10
If diabetes indeed is associated with higher mortality after infection, underlying mechanisms are unknown. Several mechanisms could explain these survival differences. For instance, animal and human models of infection suggest that immune abnormalities in diabetes, such as higher proinflammatory,11 12 procoagulant and antifibrinolytic activity,13 14 and higher expression of pathogen recognition cell surface receptors,15 16 could worsen during acute illness and increase mortality. An alternative mechanism for higher mortality among those with diabetes is increased risk of acute organ dysfunction due to higher chronic disease burden.
We therefore examined two multicentre observational cohort studies to understand the effect of diabetes on the host immune response and outcomes of pneumonia. We analysed a cohort of patients with community-acquired pneumonia (CAP) enrolled in the Genetic and Inflammatory Markers of Sepsis (GenIMS) study, and the subgroup of the population-based cohort, Health, Ageing, and Body Composition (Health ABC) study, who required hospitalisation. In both cohorts, we tested the hypothesis that pre-existing diabetes is associated with increased mortality within the first year, independent of pre-existing chronic diseases. We then determined whether higher mortality is attributable to CAP per se by comparing survival differences over the first year in Health ABC between CAP and non-infectious hospitalisations. Finally, in GenIMS we tested the hypothesis that survival differences between those with and without diabetes were due to differences in immune response and a higher risk of acute organ dysfunction during infection.
Methods
Subjects and design
We analysed subjects enrolled in the GenIMS cohort to assess differences in mortality, organ dysfunction and immune response. GenIMS is a prospective multicentre observational cohort of subjects with CAP enrolled in emergency departments (EDs) of 28 academic and community hospitals in four US regions—that is, southwestern Pennsylvania, Connecticut, southern Michigan and western Tennessee. Eligibility criteria are shown in table 1. Of the 2320 subjects enrolled, we excluded 288 patients because they were discharged from the ED and an additional 137 patients because the clinical team ruled out CAP during the first 3 days of hospitalisation, thus restricting the analysis to the remaining 1895 subjects.
Characteristics, eligibility criteria and methods to determine clinical and outcome measures in Genetic and Inflammatory Markers of Sepsis (GenIMS) and Health, Ageing and Body Composition (Health ABC) cohorts
To assess whether higher mortality is attributable to CAP, we analysed subjects enrolled in the Health ABC study, a population-based observational cohort of 70- to 79-year-old well-functioning participants. Health ABC participants were enrolled from the same geographic regions as GenIMS, including southwestern Pennsylvania (Pittsburgh) and western Tennessee (Memphis). Of the 3075 enrolled, we analysed 1645 (53.5%) subjects who were hospitalised at least once during the first 5 years of follow-up. We excluded an additional six subjects whose diabetes status was not known, restricting the analysis to the remaining 1639 subjects. Methods to ascertain hospitalisation have been described previously.20 Briefly, we assessed outcomes after the first major hospitalisation following enrolment, comparing mortality rate within the first year among those initially hospitalised for CAP and non-infectious illnesses (see appendix 1). For each cohort, the Institutional Review Boards at each site approved the study. Informed consent was obtained from participants or next of kin for GenIMS and from participants for Health ABC.
Outcome and clinical variables
For GenIMS and Health ABC cohorts, the primary outcome variable was all-cause mortality rate within the first year (table 1). We examined several additional secondary outcome variables in GenIMS. We examined cause-specific mortality using National Death Index codes. The validity of this method has been described previously.21 22 To compare risk of developing acute organ dysfunction during hospitalisation, we compared risk of severe sepsis (infection plus organ dysfunction) using Consensus criteria,17 and the risk of individual organ dysfunction for six organ systems. For acute kidney injury, we used the RIFLE criteria.23 The RIFLE criteria classifies acute kidney injury into three categories of severity using changes in serum creatinine and urine output (Risk, Injury, and Failure corresponding to mild, moderate, and severe kidney injury, respectively). Finally, we used measures of severity of illness at hospital presentation, including APACHE III (Acute Physiology and Chronic Health Evaluation III) and the Pneumonia Severity Index (PSI).24 25 Details of methods to assess co-morbid conditions in both cohorts, including diabetes, are included in table 1. Microbiological characteristics were available only in GenIMS and were assessed using blood and sputum cultures (see Online repository).
Laboratory procedures
We assessed differences in the immune responses to infection in GenIMS during hospitalisation between those with and without diabetes by comparing changes in circulating concentrations of biomarkers within inflammatory (tumour necrosis factor α (TNFα), interleukin 6 (IL-6), IL-10), coagulation (Factor IX, thrombin–antithrombin complexes (TATs), antithrombin) and fibrinolysis (plasminogen activator inhibitor-1 (PAI-1) and D-dimer) systems, and expression of cell surface markers (CD120a and CD120b (signalling receptors for TNF), human leucocyte antigen (HLA)-DR and Toll-like receptor2 (TLR2) and TLR4) on presentation to the ED and over the first week. Details of sample collection and processing have been described previously.26
We used an automated chemiluminescent immunoassay analyser (IMMULITE, Diagnostic Products, Los Angeles, California, USA) to analyse TNF, IL-6 and IL-10. We analysed coagulation and fibrinolysis markers in a random subset of 734 subjects by a commercial laboratory (Esoterix, Agoura Hills, California, USA). Specific methods and kits used were: D-dimer, latex immunoassay (Diagnostica Stago, Parsippany, New Jersey, USA); PAI-1, bio immunoassay (Biopool Chromolize, Biopool International, Ventura, California, USA); antithrombin, chromogenic (BioMerieux, Rhône-Alpes, France); Factor IX, clot (BioMerieux); and TAT, ELISA (Behring, King of Prussia, Pennsylvania, USA).
We analysed cell surface markers on ED presentation and on the third and seventh day. We analysed cell surface markers in a subset of 624 subjects enrolled in hospitals located within 60 miles of the University of Pittsburgh because samples for cell surface markers have to be analysed within 48 h. We obtained fluorochrome- or biotin-conjugated antibodies from eBioScience (San Diego, California, USA) for TLR2, TLR4 and HLA-DR, and from Invitrogen (Carlsbad, California, USA) for CD120a and CD120b. We incubated these antibodies with whole blood, lysed the red blood cells and then washed, fixed and stored the remaining cells at −4°C. We acquired cell surface marker data within 48 h of fixation on a BD FACSVantage SE flow cytometer (San Jose, California, USA) and used BD CellQuest software. Additional details of the assays for all markers are included in the Online repository.
Statistical analyses
We conducted univariate comparisons of clinical characteristics for subjects with and without diabetes using χ2 and Student t test and their non-parametric counterparts when necessary in both cohorts. We compared the mortality rate within the first year by constructing Kaplan–Meier survival curves for subjects with and without diabetes and used the Cox proportional hazards model. For the proportional hazards model, we confirmed that the HRs were similar over different intervals. We constructed serial models, including unadjusted, adjusted for demographic characteristics (age, sex and race), and adjusted for demographic characteristics and chronic diseases associated with diabetes, such as cardiovascular disease and CKD. To assess if mortality differences were attributable to CAP, we compared mortality in those hospitalised initially for CAP and other non-infectious illnesses in Health ABC. We tested for an interaction between diabetes and the reason for initial hospitalisation and mortality rate within the first year.
We assessed mechanisms of increased mortality in GenIMS. We compared risk of severe sepsis and individual organ dysfunction using logistic regression analyses. We compared differences in immune response to infection, comparing biomarkers and cell surface markers in subjects with and without diabetes at ED presentation and during the first week of hospital stay. We used regression analysis with mixed models to account for correlation of these markers over time. We also compared inflammatory and coagulation biomarkers at hospital discharge in the subset that was discharged alive and appeared to have recovered clinically to compare resolution of the immune response. We have previously shown that those with an unresolved immune response, as evidenced by higher concentrations of biomarkers at hospital discharge, had higher mortality over the first year after hospital discharge.27 To account for biomarkers that were truncated because they were below the detection thresholds, we used Tobit models to compare biomarker concentrations.28 Due to the large number of comparisons for the cell surface markers, we reported p values adjusted for false discovery rate at 0.05.29
Results
Baseline characteristics of the GenIMS cohort
Table 2 shows pre-hospitalisation characteristics for 1895 subjects enrolled in GenIMS. The average age of the cohort was 67 years and two-thirds had a history of smoking. Respiratory disease was the most common chronic disease and occurred in approximately a third of the cohort, and cardiovascular disease occurred in a quarter of the cohort. Diabetes occurred in 384 (20.3%) subjects. The prevalence of other chronic diseases, such as CKD, cancer and HIV, was low (<5%).
Demographic and clinical characteristics of all subjects, stratified by diabetes, enrolled in the Genetic and Inflammatory Markers of Sepsis (GenIMS) and Health, Ageing and Body Composition (Health ABC) study
Subjects with diabetes, on average, were 3 years older than subjects without diabetes, but results did not reach statistical significance. Those with diabetes were more likely to have CKD (10.4% vs 3.4%, p<0.0001) and cardiovascular disease (42.9% vs 21.3%, p<0.0001). HIV was more prevalent among those without diabetes (2.4% vs 0.3%, p=0.02) and only one subject with diabetes had HIV. No differences were seen in the prevalence of smoking, cancer and respiratory disease between the two groups.
Blood or sputum cultures to determine microbiological aetiology were obtained in 1606 (84.7%) subjects within 48 h of ED presentation. An aetiological agent was identified in 186 (11.5%) subjects, and this frequency was similar among those with and without diabetes (12.6% vs 11.3%, p=0.5). Among subjects in whom an aetiological agent was identified, Gram-negative organisms were more common among those with diabetes, Gram-positive organisms were more common among those without, and no differences were seen in the frequency of mixed or anaerobic organisms (7.5% vs 3.1%, 4.2% vs 7.5%, and 1% vs 0.7% for Gram-negative, Gram-positive and mixed or anaerobic organisms, respectively, p=0.001).
Diabetes was associated with higher acute organ dysfunction in GenIMS
At ED presentation, subjects with diabetes had greater illness severity, as evidenced by higher PSI scores (111.1 vs 96.9, p<0.0001), higher frequency of subjects with diabetes in PSI classes IV and V (68.7% vs 53.6%, p<0.0001), and higher APACHE III scores (59.5 vs 55.2, p<0.0001). When points for the physiology components of the APACHE III score were compared, those with diabetes had a higher score compared with those without diabetes (42.6 vs 39.8, p<0.0001).
Subjects with diabetes had higher risk of severe sepsis compared with those without diabetes (34.6% vs 29.7%, OR 1.25, 95% CI 0.99 to 1.58), but results were not statistically significant (p=0.06). This association was not statistically significant when adjusted for age, sex, race, and pre-existing cardiovascular disease and CKD (OR 1.23, 95% CI 0.96 to 1.59, p=0.09).
Diabetes increased the risk of acute kidney injury (30.2% vs 22.9% for subjects with and without diabetes, p=0.007) and this association remained significant when adjusted for age, sex and race (p=0.02). The association persisted when subjects with CKD were excluded (29.3% vs 23.6%, p=0.05). The higher risk of acute kidney injury was mainly due to increased risk of moderate (RIFLE-I) kidney injury (7.6% vs 3.6%), and only small differences were observed in the risk of mild (RIFLE-R) (10.9% vs 9.6%) and severe kidney injury (RIFLE-F) (11.6% vs 9.6%). No differences were observed in the risk of respiratory, neurological, cardiovascular, coagulation and liver dysfunction (table 3). Furthermore, those with and without diabetes were equally likely to require mechanical ventilation (6.8% vs 7%, p=0.87) and intensive care unit (ICU) admission (14.1% vs 16.5%, p=0.25), and only a small difference was seen in the median length of hospital stay (7.8 vs 7.4 days, p=0.02).
Risk of severe sepsis and organ dysfunction among subjects with and without diabetes*
Diabetes was associated with higher mortality in GenIMS
Subjects with diabetes had higher mortality rates over the first year (unadjusted HR was 1.41, 95% CI 1.12 to 1.76, p=0.002; figure 1 and table 4). The HR remained unchanged and the association remained statistically significant when adjusted for demographics (HR 1.32, 95% CI 1.05 to 1.66, p=0.01) and additionally adjusting for higher burden of cardiovascular disease and CKD (HR 1.3, 95% CI 1.03 to 1.65, p=0.02).
Kaplan-Meier survival curves over 1 year showing higher risk of death for subjects with diabetes compared with those without diabetes following hospitalisation for community-acquired pneumonia in GenIMS.
Unadjusted and adjusted HRs with 95% CIs for mortality over 1 year for subjects with diabetes compared with those without using Cox proportional hazards model
The mortality at 1 year in individuals who developed acute kidney injury was higher compared with those who did not develop it, in both subjects with and without diabetes, but results did not reach statistical significance among subjects with diabetes (30.7% vs 21.4%, p=0.08 and 28.7% vs 14.3%, p<0.0001 in subjects with and without acute kidney injury stratified by presence and absence of diabetes, respectively).
Causes of death were different in those with and without diabetes (p=0.03). Deaths due to cardiovascular disease and kidney disease were higher among those with diabetes (34.4% vs 26.8% and 10.4% vs 4.5% for cardiovascular and kidney disease, respectively). Deaths due to cancer were lower among those with diabetes (12.5% vs 26.5%), but no differences were observed in deaths due to infection, chronic respiratory disease and other causes (15.6% vs 15%, 16.7% vs 14.7% and 10.4% vs 12.5% among those with and without diabetes for infection, chronic respiratory disease and other causes, respectively).
Diabetes was associated with higher mortality after pneumonia compared with non-infectious illnesses in Health ABC
The clinical characteristics of subjects in Health ABC at enrolment are shown in table 2. The average age was 73 years and approximately half were whites and females. The prevalence of diabetes was similar in Health ABC and GenIMS (n=299, 18.2% and n=384, 20.3% in Health ABC and GenIMS cohorts, respectively). Compared with GenIMS, the prevalence of respiratory disease was lower in Health ABC (24.2% vs 37.9%), whereas CKD was more common in Health ABC (24.5% vs 9.8%). Similar to GenIMS, subjects with diabetes enrolled in Health ABC had a higher prevalence of CKD (34.1% vs 22.4%, p<0.0001) and cardiovascular disease (35.9% vs 25.9%, p=0.03).
An interaction was observed between diabetes and the reason for the initial hospitalisation and mortality within the first year. In the unadjusted model, pre-existing diabetes was associated with higher risk of death after hospitalisation for CAP compared with those hospitalised for non-infectious illnesses (HR 1.87, 95% CI 0.76 to 4.6, and 1.16, 95% CI 0.8 to 1.6 for those hospitalised for CAP and non-infectious illnesses, respectively; interaction p=0.04 (table 4)). This interaction persisted in models adjusted for demographics (HR 1.82, 95% CI 0.72 to 4.62, and 1.08, 95% CI 0.76 to 1.52 for those hospitalised for CAP and non-infectious illnesses, respectively; interaction p=0.04) and additionally adjusted for pre-existing cardiovascular disease and CKD (HR 1.8, 95% CI 0.66 to 4.92, and 1.06, 95% CI 0.75 to 1.51 for those hospitalised for CAP and non-infectious illnesses, respecteively; interaction p=0.09). Forty-one participants who were hospitalised initially for non-infectious illnesses were subsequently hospitalised for infections. Excluding these subjects did not alter the results, and an interaction between diabetes, reason for hospitalisation and mortality within the first year persisted (unadjusted HR 1.87, 95% CI 0.76 to 4.60, and 0.97, 95% CI 0.66 to 1.41 for those hospitalised for CAP and non-infectious illnesses, respectively; interaction p=0.01).
Diabetes did not alter immune response after CAP hospitalisation
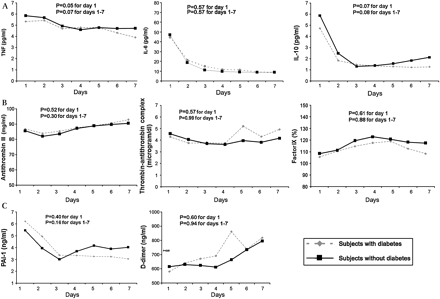
In GenIMS, the circulating concentrations of inflammatory (TNF, IL-6 and IL-10), coagulation (antithrombin, Factor IX and TAT complexes) and fibrinolysis (PAI-1 and D-dimer) biomarkers were similar among subjects with and without diabetes at ED presentation and over the first week of hospitalisation (figure 2). No differences were seen in expression of CD120a, CD120b, HLA-DR, TLR4 and TLR2 on monocytes between those with and without diabetes (figure 3). Expression of TLR2 on granulocytes was higher among subjects with diabetes on day 1 and 3, but higher among those without diabetes on day 7 (p=0.04). At hospital discharge, those with diabetes had higher IL-6 concentrations compared with those without, but these differences were small (8.67 vs 7.41 pg/ml, p=0.03) (table 5). No differences were observed in other biomarkers at hospital discharge.
No differences were observed in geometric means of inflammatory (tumour necrosis factor, interleukin 6 (IL-6) and IL-10 shown in A), coagulation (Factor IX, antithrombin and thrombin–antithrombin complexes shown in B) and fibrinolysis (plasminogen activator-1 PAI-1) and D-dimer shown in C) biomarkers in 1427, 734 and 734 subjects with and without diabetes, respectively. Biomarkers were measured on presentation to the emergency department and over the first week of hospitalisation, and p values are shown for these comparisons.
No differences were observed in cell surface markers on presentation to the emergency department and on the third and seventh day in 624 subjects with and without diabetes. Values for cell surface markers were reported as the mean channel fluorescence of cells positive for a given cell surface marker. p Values are shown for comparisons over time. Only expression of Toll receptor 2 (TLR2) on granulocytes appeared to be higher among subjects with diabetes on day 1 and 3 and were higher in those without diabetes on day 7 (p=0.04).
Association between inflammatory, coagulation, fibrinolysis markers (geometric means with SD) at hospital discharge stratified by diabetes among 1808 subjects discharged alive from the hospital
Plasma glucose concentrations
Plasma glucose was available at ED presentation in most GenIMS subjects (n=1795, 94.7%). Those with diabetes had higher median plasma glucose compared with those without diabetes (190 mg/dl, IQR 136–256 mg/dl vs 117 mg/dl, IQR 99–143 mg/dl, p<0.0001).
Regardless of diabetes, non-survivors at 1 year had higher median blood glucose on ED presentation compared with survivors, but differences were small (130 mg/dl, IQR 103–177 mg/dl vs 122 mg/dl, IQR 102–158 mg/dl, p=0.02). Hyperglycaemia at ED presentation (glucose >200 mg/dl) was associated with higher mortality within the first year (HR 1.31, 95% CI 1.01 to 1.69, p=0.03). However, results did not reach statistical significance when adjusted for age, sex and race (HR 1.27, 95% CI 0.98 to 1.64, p=0.06). Furthermore, the HRs were different when the analyses were stratified by presence or absence of diabetes (HR 0.89, 95% CI 0.6 to 1.32, p=0.57 in subjects with diabetes and HR 1.4, 95% CI 0.98 to 2.11, p=0.06 in subjects without diabetes).
Discussion
We found that diabetes was common and present in 20% of subjects with CAP. Compared with subjects without diabetes, those with diabetes had a higher mortality within the first year after CAP and a quarter died at 1 year. The higher mortality was not confounded by higher burden of pre-existing cardiovascular disease and CKD. Furthermore, diabetes was associated with higher mortality following hospitalisation for CAP compared with hospitalisation for non-infectious illnesses. These results suggest that higher mortality within the first year among individuals with diabetes is attributable to the pneumonia hospitalisation. Diabetes did not modify the immune response in a broad panel of circulating inflammatory, coagulation, fibrinolysis and cell surface markers, suggesting that differences in immune response are unlikely to explain survival differences. Based on patterns of organ dysfunction observed during the hospital course and cause-specific mortality, the higher incidence of acute kidney injury and acceleration of underlying cardiovascular disease may mediate higher mortality within the first year after CAP among subjects with diabetes.
Our results showing survival differences due to diabetes after CAP hospitalisation have several strengths. First, the higher hazards of death due to diabetes were similar in two cohorts, although results did not reach statistical significance in Health ABC due to the small number of CAP events. Secondly, we assessed survival differences after the first hospitalisation event in Health ABC to avoid confounding due to preceding non-infectious illnesses, such as acute myocardial infarction, which are common and may be associated with worse survival in diabetes.10 We also demonstrated an interaction between diabetes, reason for hospitalisation and mortality within the first year. Among those with diabetes, mortality was higher after CAP hospitalisation compared with hospitalisation for non-infectious illnesses. Thirdly, survival differences persisted after adjusting for differences in chronic disease burden, such as cardiovascular disease and CKD. These results suggest that the higher mortality within the first year in diabetes can be attributed to the CAP hospitalisation and cannot be explained by higher burden of pre-existing cardiovascular disease and CKD among those with diabetes.
We speculate that acceleration of pre-existing chronic disease may explain higher long-term mortality among those with diabetes. For instance, cardiovascular disease accounted for more than third of all deaths in individuals with diabetes. Pre-existing cardiovascular disease was more common among those with diabetes, which may be further accelerated by the acute infection. Early recognition or better management of atherosclerotic heart disease and concomitant risk factors, such as smoking and hyperlipidaemia, may improve outcomes. We showed that diabetes was associated with higher risk of acute kidney injury, which was associated with higher risk of 1-year mortality in our study and previous studies.30 Acute kidney injury itself or its sequela, CKD, may lead to death by several mechanisms, including increased risk of cardiovascular disease and infections.
Our results may explain the conflicting results of previous studies which assessed the association between diabetes and survival after CAP. First, mortality was assessed at different time points in earlier studies. Our results suggest that, although the RR of death was similar at different time points, statistically significant differences are more likely to be seen in studies that assessed long-term outcomes due to fewer deaths at earlier time points.3 Secondly, we examined the effect of diabetes on outcomes, conditional upon developing pneumonia. Previous population-based studies that showed higher incidence of death due to infections in subjects with diabetes could not assess whether these differences were due to increased susceptibility to infections or worse outcomes after infection.5 6
We showed a 1.3-fold higher risk of acute kidney injury among subjects with diabetes, even among subjects without a history of CKD. Mechanisms underlying increased risk of acute kidney injury after CAP among those with diabetes are unclear. We speculate that the higher risk of acute kidney injury in subjects with diabetes could be due to higher prevalence of subclinical kidney disease prior to pneumonia or due to higher risk of developing contrast-induced nephropathy.31 32
Although immune dysfunction in diabetes is well recognised, the lack of a clear influence of diabetes on a broad panel of biomarkers and cel surface markers at multiple time points during hospitalisation for CAP is remarkable. We observed differences in expression of TLR2 on granulocytes over time between subjects with and without diabetes, but the direction of this difference was not consistent over time and was uncertain in terms of biological significance. We chose inflammatory, coagulation and fibrinolysis biomarkers because these biomarkers are altered in diabetes11 33 and in human endotoxaemia models by hyperinsulinaemia and hyperglycaemia.12 14 We chose TLR2 and TLR4 because expression of these cell surface markers and their downstream mediators is upregulated in diabetes16 and they play an important role in host response to infection and sepsis.15 We performed serial measurements of these markers, including on presentation to the ED, when immune responses are least likely to be modified by therapeutic interventions. It is likely that diabetes may modify these markers prior to ED presentation, or other mechanisms could be influenced by diabetes, such as alterations in neutrophil function, apoptosis, oxidative stress and chemokines. However, the lack of effect of diabetes on the immune response in a broad panel of circulating biomarkers and cell surface markers suggest that pre-existing diabetes does not influence the immediate host response to CAP, possibly because the responses elicited by pneumonia are much more profound than the relatively modest alterations produced by diabetes per se.
Our study has limitations. First, we assessed the effect of hyperglycaemia, a potential mechanism of increased mortality, on ED presentation only, and serial glucose levels were not available. Secondly, we used different criteria to diagnose diabetes in both cohorts. Stringent criteria were used in Health ABC, including self-report and review of medication inventory, and fasting blood glucose in subjects who did not report diabetes. In GenIMS, we used a combination of self-report and review of medication inventory, and we may have misclassified some individuals as non-diabetic. The association between admission hyperglycaemia and higher mortality only among those without diabetes in this cohort suggests that such a misclassification bias would skew the results towards the null and attenuate the HRs between diabetes and long-term survival. Indeed, the HRs were slightly lower in GenIMS, suggesting that such a misclassification bias is unlikely to negate our results. Finally, we could not assess whether our results were confounded due to differences in microbiological aetiology between those with and without diabetes. Although cultures were obtained in most subjects, we identified an aetiological agent in a small subgroup. The low yield of cultures in our study is consistent with previous large studies of patients with CAP34 35 and probably due to poor yield of current culture techniques. Larger studies will be necessary to understand differences in immune response for different aetiological agents.
In summary, once CAP occurs, those with diabetes were more likely to die over 1 year. The mechanism is unlikely to be due to alterations in immune response, at least as measured by a broad panel of circulating biomarkers and cell surface markers. The higher mortality may be due to worsening of pre-existing cardiovascular disease or higher risk of acute kidney injury.
Acknowledgments
We are indebted to the nurses, respiratory therapists, phlebotomists, physicians and other healthcare professionals who participated in GenIMS, as well as the subjects and their families who supported GenIMS and the Health ABC study. A complete list of GenIMS investigators is available at http://www.ccm.upmc.edu/genims_investigators
Appendix 1 List of non-infectious causes of hospitalisation in Health ABC study
Myocardial infarction
Angina/other ischaemic disease
Congestive heart failure
Carotid artery disease
Peripheral arterial disease
Stroke or cerebrovascular accident (CVA)
Transient ischaemic attack
Chronic obstructive pulmonary disease/emphysema/asthma
Upper gastrointestinal bleeding
Lower gastrointestinal bleeding
Abdominal hernia
Benign prostatic hyperplasia
Gallbladder disease
Cancer
Depression
Dementia
Osteoarthritis
Fracture
Neoplasms
Endocrine, nutrition, metabolic diseases
Diseases of blood and blood-forming organs
Mental disorders (not dementia and depression)
Diseases of nervous system, other than stroke
Diseases of circulatory system, other than myocardial infarction, angina and congestive heart failure
Diseases of respiratory system, other than chronic obstructive pulmonary disease, asthma, emphysema
Diseases of digestive system, other than bleeding
Diseases of genitourinary system, other than benign prostatic hyperplasia
Diseases of skin
Diseases of musculoskeletal and connective tissue, other than osteoarthritis
Ill-defined symptoms and signs
Injury and poisoning, excluding fractures
References
Supplementary materials
Web Only Data thx.2010.136317
Files in this Data Supplement:
Footnotes
Funding GenIMS was funded by NIGMS R01 GM61992 with additional support from GlaxoSmithKline for enrolment and clinical data collection, and Diagnostic Products Corporation for the cytokine assays. Health ABC was funded by NIA (N01-AG-6-2101, N01-AG-6-2103, N01-AG-6-2106), NHLBI (R01HL74104) and NIAID (27913 and 39482). Health ABC was supported in part by the Intramural Research Program of the NIH, National Institute on Ageing. SY is supported by K23GM083215.
Competing interests None.
Ethics approval This study was conducted with the approval of the ethics committee at each participating site.
Provenance and peer review Not commissioned; externally peer reviewed.