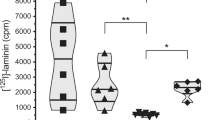
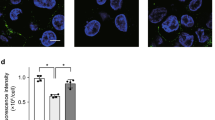
Abstract
In the lungs of cystic fibrosis patients, overproduction of mucus leads to morbidity and mortality by obstructing airflow and shielding bacteria from antibiotics. Here we demonstrate that overproduction of mucus is a direct result of the activation of mucin gene expression by Gram-positive bacteria. Bacterial lipoteichoic acid activates the platelet-activating factor receptor, which is G protein–coupled. This results in activation of a disintegrin and metalloproteinase (ADAM10), kuzbanian, cleavage of pro heparin–binding epidermal growth factor and activation of the epidermal growth factor receptor. Unlike responses in macrophages, the epithelial-cell response to lipoteichoic acid does not require Toll-like receptor 2 or 4.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Li, J. -D. et al. Transcriptional activation of mucin by P. aeruginosa lipopolysaccharide in the pathogenesis of cystic fibrosis lung disease. Proc. Natl. Acad. Sci. USA 94, 967–972 (1997).
Dohrman, A. et al. Mucin (MUC 2 and MUC 5 AC) transcriptional regulation in response to Gram-positive and -negative bacteria. Biochim. Biophys. Acta 1406, 251–259 (1998).
Li, J.-D. et al. Activation of NFkB via a Src-dependent Ras-MAPK-pp90rsk pathway is required for P. aeruginosa-induced mucin overproduction in epithelial cells. Proc. Natl. Acad. Sci. USA 95, 5718–5723 (1998).
Boat, T. & Boucher, R. in Textbook of Respiratory Medicine (eds. Murray, J. & Nadel, J.) 1418–1450 (W.B. Saunders, Philadelphia, 1994).
Taddei, F. et al. Role of mutator alleles in adaptive evolution. Nature 387, 700–702 (1997).
Oliver, A., Canton, R., Campo, P., Baquero, F. & Blazquez, J. High frequency of hypermutable Pseudomonas aeruginosa in cystic fibrosis lung infection. Science 288, 1251–1254 (2000).
Finkbeiner, W.E. & Basbaum, C.B. Monoclonal antibodies directed against human airway secretions. Am. J. Pathol. 131, 290–297 (1988).
Kuan, S.F., Basbaum, C., Byrd, J. & Kim, Y. Characterization of high and low mucin variants from colon tumor cell line LS174T. Cancer Res. 47, 5715–5724 (1987).
Gum, J., Hicks, J. & Kim, Y. Identification and characterization of the MUC 2 (human intestinal mucin) gene 5′ flanking region: promoter activity in cultured cells. Biochem. J. 325, 259–267 (1997).
Cundell, D., Gerard, N., Gerard, C., Idanpaan-Heikkila, I. & Tuomanen, E. Streptococcus pneumoniae anchor to activated human cells by the receptor for platelet-activating factor. Nature 377, 435–438 (1995).
Cundell, D., Gerard, C., Idanpaan-Heikkala, I., Tuomanen, E. & Gerard, N. PAF receptor anchors Streptococcus pneumoniae to activated human endothelial cells. Adv. Exp. Med. Biol. 416, 89–94 (1996).
Daub, H., Weiss, F., Wallasch, C. & Ullrich, A. Role of transactivation of the EGF receptor in signaling by G-protein-coupled receptors. Nature 379, 557–560 (1996).
Prenzel, N. et al. EGF receptor transactivation by G-protein-coupled receptors requires metalloproteinase cleavage of proHB-EGF. Nature 402, 884–888 (1999).
Maudsley, S. et al. The β2-adrenergic receptor mediates extracellular signal-regulated kinase activation via assembly of a multi-receptor complex with the epidermal growth factor receptor. J. Biol. Chem. 275, 9572–9580 (2000).
Aderem, A. & Ulevitch, R. Toll-like receptors in the induction of the innate immune response. Nature 406, 782–787 (2000).
Schwandner, R., Dziarski, R., Wesche, H., Rothe, M. & Kirschning, C. Peptidoglycan and lipoteichoic acid-induced cell activation is mediated by toll-like receptor 2. J. Biol. Chem. 274, 17406–17409 (1999).
Takeuchi, O. et al. Differential roles of TLR2 and TLR4 in recognition of Gram-negative and Gram-positive bacterial cell wall components. Immunity 11, 443–451 (1999).
Michelsen, K. et al. Role of Toll-like receptors in bacteria-induced maturation of murine dendritic cells. J. Biol. Chem. 276, 25680–25686 (2001).
Opitz, B. et al. Toll-like receptor 2 mediates Treponema glycolipid and lipoteichoic acid-induced NFkB translocation. J. Biol. Chem. 276, 22041–22047 (2001).
Medzhitov, R. et al. MyD88 is an adaptor protein in the hToll/IL-1 receptor family signaling pathways. Mol. Cell 2, 253–258 (1998).
Mograbi, B. et al. Human monocytes express amphiregulin and heregulin growth factors upon activation. Eur. Cytokine Netw. 8, 73–81 (1997).
Hardie, W. et al. Immunolocalization of transforming growth factor alpha and epidermal growth factor receptor in lungs of patients with cystic fibrosis. Pediatr. Dev. Pathol. 2, 415–423 (1999).
Stahlman, M.T., Gray, M.E., Chytil, F. & Sundell, H. Effect of retinol on fetal lamb tracheal epithelium, with and without epidermal growth factor. Lab. Invest. 59, 25–35 (1988).
St. George, J.A. et al. Effect of epidermal growth factor on the fetal development of the tracheobronchial secretory apparatus in Rhesus monkey. Amer. J. Respir. Cell Mol. Biol. 4, 95–101 (1991).
Guzman, K., Randell, S. & Nettesheim, P. Epidermal growth factor regulates expression of the mucous phenotype of rat tracheal epithelial cells. Biochem. Biophys. Res. Commun. 217, 412–418 (1995).
Takeyama, K. et al. Epidermal growth factor system regulates mucin production in airways. Proc. Natl. Acad. Soc. USA 96, 3081–3086 (1999).
Albert, D. et al. ABT-299, a novel PAF antagonist, attentuates multiple effects of endotoxemia in conscious rats. Shock 6, 112–117 (1996).
Ishii, I., Saito, E., Izumi, T., Ui, M. & Shimizu, T. Agonist-induced sequestration, recycling and resensitization of platelet activating factor receptor. J. Biol. Chem. 273, 9878–9885 (1998).
Trapaidze, N., Keith, D., Cvejic, S., Evans, C. & Devi, L. Sequestration of the delta opioid receptor. Role of the C terminus in agonist-mediated internalization. J. Biol. Chem. 271, 29279–29285 (1996).
Gum, J. et al. The human MUC 2 intestinal mucin has cysteine-rich subdomains located both upstream and downstream of its central repetitive region. J. Biol. Chem. 267, 21375–27383 (1992).
Acknowledgements
We thank R. Medzhitov for providing the Myd88 mutant MyDC; Z. Werb and D. Julius for discussion; M. Gallup, E. Drori and S. Sidhu for collaboration. This work was supported by NIH grants HL43726 and HL 24136 as well as by a grant from the Cystic Fibrosis Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lemjabbar, H., Basbaum, C. Platelet-activating factor receptor and ADAM10 mediate responses to Staphylococcus aureus in epithelial cells. Nat Med 8, 41–46 (2002). https://doi.org/10.1038/nm0102-41
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0102-41
This article is cited by
-
ERdj5 protects goblet cells from endoplasmic reticulum stress-mediated apoptosis under inflammatory conditions
Experimental & Molecular Medicine (2023)
-
On the role of bacterial metalloproteases in COVID-19 associated cytokine storm
Cell Communication and Signaling (2021)
-
Dietary Bacillus spp. enhanced growth and disease resistance of weaned pigs by modulating intestinal microbiota and systemic immunity
Journal of Animal Science and Biotechnology (2020)
-
TNFα promotes mucosal wound repair through enhanced platelet activating factor receptor signaling in the epithelium
Mucosal Immunology (2019)
-
The association of the expression of miR-122-5p and its target ADAM10 with human breast cancer
Molecular Biology Reports (2015)