Abstract
Severe spinal collapse and pulmonary function deterioration are so common in patients with Duchenne muscular dystrophy (DMD). The limit for scoliosis surgery has been a minimum forced vital capacity (FVC) of about 30% of predicted value. DMD patients with low %FVC who might benefit from scoliosis surgery have not been being offered surgery. Between 2005 and 2007, a total of 17 DMD patients with FVC of <30% at admission underwent scoliosis surgery. %FVC at admission was 22% (range 16–27%). After admission, they were trained with inspiratory muscle training, using a pulmonary trainer (threshold IMT) for 6 weeks prior to surgery and encouraged to continue the training even after surgery and discharge. %FVC increased in all patients and noted 26% (range 22–31%) the day before surgery. The preoperative scoliosis was 97° (range 81°–130°). All patients had posterior spinal fusion and were extubated on the operative day. No patients developed any respiratory complications. No ventilatory support was needed. The mean ICU stay was 0.5 days (range 0–1 day). The postoperative scoliosis was 31° (range 18°–40°). DMD patients with severe scoliosis and FVC considered too low to permit reasonable surgical risk could undergo surgery after inspiratory muscle training, with no major complications.
Similar content being viewed by others
Introduction
Posterior spinal fusion and instrumentation for scoliosis in DMD has been aimed at maximizing function and improving patients’ QOL [2, 4–9, 16–18, 20, 26–30]. This surgery has been widely accepted as an optimal procedure to prevent progression of scoliosis and maintain up-right and comfortable sitting balance [2, 4–9, 16–18, 20, 26–30]. Despite the magnitude of the surgery, most of the patients and their families have believed scoliosis surgery improves their function, cosmesis, sitting balance and QOL [6].
Scoliosis is almost universal in DMD patients. The natural history of DMD scoliosis has been well established and progressive scoliosis generally develops relentlessly to levels as great as 100° or more [21, 33, 36]. Scoliosis surgery should be early enough in the course of the underlying disease, when cardiac and pulmonary function is sufficient, so that patients can be anesthetized and operated relatively safely and in order to reduce the likelihood of major complications [2, 5, 9, 36]. Irreversible changes in cardiac and pulmonary function pose serious risks to anesthesia and have been relative contraindications to surgery. A positive correlation between decreased pulmonary function and increased pulmonary complications has been reported [9, 21, 28, 30, 33, 36]. Some authors reported that the mean forced vital capacity (FVC) dropped to <40% of predicted value when the mean scoliotic curve measured only 23° [24]. Many authors have shown if a patient has a FVC of <35% of predicted value, prolonged ventilatory support is usually required, therefore, conclude scoliosis surgery is recommended only for a patient who has a FVC of at least 35–40% [24, 27, 31, 35].
Thus, the limit for scoliosis surgery has been a minimum FVC of about 30–35% of predicted value [9, 17, 21, 24, 25, 27, 31, 35, 36] and such patients with poor pulmonary function who might benefit from scoliosis surgery have not been being offered scoliosis surgery.
Recently, several authors reported such patients with severely compromised pulmonary function (FVC < 30%) on noninvasive mechanical ventilation could be safely operated for scoliosis surgery [3, 9]. However, extubations are delayed and mechanical ventilatory support is often prolonged [3, 9]. Prolonged ICU care is also usually required.
Pulmonary function deterioration in DMD patients is due primary to inspiratory muscle weakness. Some studies have shown inspiratory muscles and diaphragm of DMD patients are trainable in terms of strength and endurance [1, 13, 15, 18, 32, 34].
Our patients with FVC conventionally considered too low to permit reasonable surgical risk have been managed successfully with no pulmonary complications after inspiratory muscle training. We present the results of scoliosis surgery in a group of patients with DMD and poor pulmonary function (FVC < 30%) not on ventilatory support. These patients with progressive scoliosis and poor pulmonary function represent an anesthetic and surgical challenge. Over the last 5 years, we have not had any DMD patient undergoing scoliosis surgery require reintubation or tracheotomy using this method.
Materials and methods
This study was approved by the Institutional Review Board of Kitasato University and informed consent was obtained from all patients prior to participation in the study.
A total of 17 consecutive patients with DMD undergoing scoliosis surgery between 2005 and 2007 were prospectively collected and followed. All the patients had standard posterior spinal fusion and instrumentation performed by the same surgeon (M.T.). Minimum 2-year follow-up was required for inclusion in this study. Inclusion criteria required that each patient who had a FVC of <30% of predicted value was (1) able to ventilate his lung autonomously; (2) not on ventilatory support, and (3) with considerable difficulties in sitting, back pain or breakdown of the skin due to scoliosis. We are of the opinion that those patients with difficulties in sitting, back pain or breakdown of the skin due to scoliosis can benefit from scoliosis surgery despite the development of severe scoliosis and significant deterioration of pulmonary function if surgery is performed successfully.
Pulmonary function tests
Pulmonary function tests were performed with a computerized pulmonary function system (FUDAC-50, Fukuda Denshi, Tokyo). The various measurements of pulmonary function were assessed. The tests were performed on each occasion with the patients seated in wheel-chair. Arm-span measurements were selected for calculations for the predicted heights. Pulmonary function value reported in this study is FVC expressed as percent of predicted value (%FVC) on the published normative data. Pulmonary function tests was evaluated in all the patients included in this study at 6 weeks and 3 weeks before surgery, the day before surgery, at 6 weeks, at 1 and 2 years after surgery and at the last follow-up. The 6-month follow-up period after surgery was chosen to avoid possible negative influences of this type of surgery (e.g. pain). %FVC was measured three times and evaluated by one independent observer. The best trial for the test was used for further analysis.
Inspiratory muscle training
After admission, patients had to perform both resistive breathing maneuvers and maximal static inspiratory efforts. They were trained with preoperative inspiratory muscle training by a pulmonary trainer (threshold IMT) (Figs. 1, 2) for 6 weeks prior to surgery. Training was performed with the patient in the sitting position and a nose was usually used. They were also encouraged to continue after surgery and even in the home after discharge. The pulmonary trainer resistive exercise intervention, presented in Table 1, was based on previously published protocols [14]. Inspiratory muscle training was performed daily for 6 weeks. Three sets of 15 repetitions must have been performed. Initial resistance (H2O cm) was set at 30% of the patient’s preset maximal inspiratory pressure (MIP). Inspiratory muscle training pressure resistance was progressed weekly according to the patient’s baseline MIP and RPE as well as the patient’s symptoms. Patients ranked perception of fatigue during training using a Borg Rating of Perceived Exertion (RPE) Scale [13]. Patients are completed a log documentation exercise adherence, Borg RPE, and symptoms following the daily exercise sessions. All the patients tolerated the exercise training program except for one subject who complained of light-headedness during the initial training session. When the light-headedness occurs, pressure resistance was reduced by 2 cm H2O.
Pulmonary trainer (threshold IMT, Philips Respironics, Inc., Murrysville, PA, USA) provides consistent and specific pressure for inspiratory muscle strength and endurance training, regardless of how quickly or slowly patients breathe. This device incorporates a flow-independent one-way valve to ensure consistent resistance and features an adjustable specific pressure setting (in cm H2O) to be set by a healthcare professional. When patients inhale through threshold IMT, a spring-loaded valve provides a resistance that exercises respiratory muscles through conditioning
Radiographic assessments
Radiographic assessments were performed on sitting anteroposterior and lateral spinal radiographs covering the whole spinal column with the patient in sitting position. These assessments were made the day before surgery, in the postoperative week, at 1 and 2 years after surgery. The Cobb angles of the curves on the coronal plane, thoracic kyphosis between T3 and T12, and lumbar lordosis between L1 and L5 on the sagittal plane were measured.
Surgical procedure
The primary aim of this surgery was to obtain a solid fusion, level pelvis and a balanced spine. All patients had posterior spine fusion and all-screw construct from the upper thoracic spine (T3 or T4) to the lower lumber spine (L5) for scoliosis. No anterior surgery was performed in any of this consecutive series. The rods were contoured to create sagittal profile and coronal balance was achieved by sequential reduction of the segments toward the rods. Autograft bone was obtained from spinous process, laminae and transverse process. The posterior elements were decorticated. Autograft bone was packed onto the prepared surfaces and placed carefully in each facet. Autotransfusion via both preoperative storage and intraoperative collection was used. During surgery, monitoring of spinal cord function was conducted by somato-sensory/motor-evoked potentials. The wound was sutured in three layers with two drainage tubes.
Peri/postoperative management
Patients were extubated in the operation room, when the ability to ventilate their lungs autonomously was documented. Intensive care unit (ICU) care was not always needed but sometimes needed only on the operative day. All patients received supplemental oxygen on the operative day weaned off with monitoring of pulse oximetry as a feedback to maintain adequate alveolar ventilation. Pulse oximetry monitoring was used to evaluate pulmonary function for all patients in the immediate postoperative period. Alveolar ventilation was considered adequate when baseline oxyhemoglobine saturation (SpO2) remained normal (SpO2 > 94%). Frequent expulsion of debris was performed by nurses in the immediate postoperative period. Sudden decreases in SpO2 should be assumed to be due to airway mucous encumberment and should be reversed by using mechanically assisted coughing (MAC). MAC has been used to increase cough flows to clear airway secretions during intercurrent respiratory tract infections. The patients were encouraged to sit up on bed on the operative day and sit in the wheelchair on postoperative day 1, with discharge occurring on postoperative day 14. The patients were encouraged to continue inspiratory muscle training after surgery and even in the home after discharge.
Results
A total of 17 consecutive patients were prospectively enrolled into this study. No patient was lost to follow-up. Demographic details and surgical parameters of all the patients are shown in Table 2. Radiographic parameters for the coronal and sagittal planes are shown in Table 3. Changes in %FVC is shown in Fig. 3. The mean %FVC at the time of admission was 22% (range 16–27%). %FVC increased in all patients at 3 weeks before surgery and it was 24% (range 19–28%). %FVC was 26% (range 22–31%) the day before surgery. There was significant difference in FVC values before and after inspiratory muscle training. The mean preoperative scoliotic curvature was 97° (range 81°–130°). The mean preoperative thoracic kyphosis was 9° (range 0°–25°) and lumbar lordosis was 16° (range 0°–65°).
Changes in %FVC are demonstrated. After preoperative respiratory muscle training for 6 weeks, %FVC increased in all patients. Although %FVC in 16 patients did not exceed 30% at the time of surgery, all the 17 patients underwent surgical correction of scoliosis successfully without any complications. At 6 weeks after operation, %FVC remained stable or increased slightly. %FVC decreased in all patients at 1 and 2 years after operation. The average rate of decline of %FVC was 3.2% per year after surgery
All patients in our series recovered well following the surgery with no major complications. There was a smooth transition from no spontaneous breathing to spontaneous breathing in all patients. They were successfully extubated in the operation room. No patients showed abnormal pulse oximetry monitoring values in the immediate postoperative period. No patients developed any respiratory complications including postoperative pneumonia or required reintubation or tracheotomy.
%FVC remained stable or increased slightly in all patients at 6 weeks after surgery and it was 27% (range 21–31%). There was no significant difference between the preoperative and postoperative values. However, %FVC continues to decrease with progression of the underlying disease and it was 23% (range 19–27%) at 1 year after surgery, 20% (range 16–22%) at 2 years after surgery and 17% (range 14–20%) at the last follow-up.
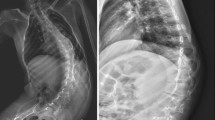
The mean immediately postoperative scoliotic curvature was 31° (range 18°–40°). The difference in the preoperative and postoperative scoliotic curvature was significant. The mean scoliotic curvature was 32° (range 18°–42°) at 1 year after surgery, 32° (range 18°–42°) at 2 years after surgery and 32° (range 18°–3°) at the last follow-up. No significant loss of correction was found between postoperative time and the last follow-up. Figure 4 is a radiographic example.
a, b A 12-year-old boy with DMD. Anteroposterior radiograph show severe scoliosis of 90°. %FVC at the time of admission was 22% and %FVC increased to 26% the day before surgery. c, d Surgical correction of scoliosis was performed successfully. Successful extubation was performed on the operative day. Postoperative sitting views show significant coronal correction of 18°, with normalization of sagittal plane. At 6 weeks after surgery, %FVC remained stable and it was 26%
The mean immediately postoperative thoracic kyphosis was 16° (range 10°–27°) and lumbar lordosis was 42° (range 30°–53°). The mean thoracic kyphosis at 1 year after surgery was 17° (range 12°–26°) and lumbar lordosis was 44° (range 30°–52°). The mean thoracic kyphosis at 2 years after surgery was 17° (range 10°–25°) and lumbar lordosis was 43° (range 33°–50°).
Ten minor complications occurred immediately after surgery. The most common complication was paralytic ileus, occurring in 6 patients. This was treated with observation without oral intake and cleared in 48 h. Transient tachycardia, which was due possibly to the presence of profound cardiomyopathy, occurred in 4 patients. This was treated successfully with medications and resolved uneventfully in at least 5 days. No respiratory complications such as postoperative pneumonia or prolonged respiratory dependence were found. There were no neurologic complications, infection or instrumentation failure during the study period. Up to the present, all patients are still alive at 3.5 years (range 1.0–5.5 years) after surgery.
Discussion
To the best of our knowledge, there have been little data addressing clinical outcomes and peri/postoperative managements in DMD patients with severe scoliosis and poor pulmonary function undergoing spine surgery.
Scoliosis is almost universal in DMD patients. Natural history studies on DMD demonstrated an almost invariable progression in the scoliosis with progression of the underlying disease [2, 5, 6, 11, 19, 27, 36]. Posterior spinal fusion for scoliosis in DMD should be early enough in the course of curve progression, when cardiac and pulmonary function are sufficient, so that the patients can be anesthetized and operated relatively safely and in order to reduce the likelihood of major complications 4, 7, 8, 12, 17, 20, 26–30]. Despite the magnitude of the surgery, posterior spinal fusion for scoliosis in DMD has been widely accepted as the optimal procedure to prevent progression of scoliosis and maintain seating balance and comfort [4, 7, 8, 12, 17, 20, 26–30].
However, this major surgery is accompanied by many significant complications [9, 21, 25, 26]. Significant complication rates from 44 to 62% have been reported in some studies [9, 21, 25, 26]. Due to poor pulmonary function in these patients, they are more susceptible to postoperative pneumonia, prolonged respirator dependence, respiratory failure, cardiac failure and death [2–6, 9, 15, 25–27, 30, 32, 33]. A strong association between restrictive pulmonary disease and increased pulmonary complications has been reported [12]. Also, a positive correlation between decreased FVC and increased pulmonary complications has been reported [22]. Rawlins et al. [23] found 19% pulmonary complications in pediatric patients undergoing spine surgery who had a FVC of <40% of predicted value. Several authors have shown if a patient has a FVC of <35%, prolonged ventilatory support is usually needed after surgery and recommended scoliosis surgery only for patients who have a FVC of at least 35–40% [27, 30]. Smith et al. [27] showed when scoliosis exceeded 35°, FVC was usually <40% and concluded that if the decision to perform scoliosis surgery was delayed until scoliosis progressed to 35°, the major surgery carried appreciable risks. These patients were not on ventilatory support.
Thus, surgeons are often reluctant to perform elective surgery on patients who are ventilator dependent or whose FVC do not exceed 40% of predicted value [3]. Usually, the limit for surgery is not the extent of the curvature but a minimum FVC of about 30% of predicted value [3, 9, 25, 27]. Several previous studies have attempted to quantify peri/postoperative complications in patients with neuromuscular disease and poor pulmonary function undergoing surgery for scoliosis. Sakai et al. [25] recommended preoperative tracheotomy if the FVC was <40% of predicted value in patients with DMD undergoing scoliosis surgery. Bach et al. [3] referred to the role of noninvasive ventilation and noted that all patients with neuromuscular scoliosis were extubated by the third postoperative day to noninvasive intermittent positive-pressure ventilation (IPPV) despite continuous ventilator dependence and no patient developed any postoperative respiratory complications or required a tracheotomy. Gill et al. [9] reported patients with poor pulmonary function (FVC < 30%) and preexisting respiratory failure on nocturnal noninvasive ventilatory support could be safely operated for scoliosis with no increased risk of complications. These patients were on either continuous invasive or noninvasive ventilation before and after surgery. Prolonged ICU care was also required. In our series, preoperative inspiratory muscle training for 6 weeks after admission was performed and %FVC increased in all patients. Although 16 of 17 patients had a FVC of <30% at the time of surgery, surgical correction of scoliosis was performed and all patients were extubated on the operative day. No prolonged ventilatory support was needed. ICU care was required only on the operative day. There were no respiratory complications. Since our patients have been managed successfully long-term with this method, we conclude that if the patient can ventilate his lung autonomously immediately after surgery, successful extubation in the operation room can be expected.
Several studies reported that respiratory muscle and diaphragm were trainable in terms of strength and endurance by inspiratory muscle training in patients with neuromuscular disease [17, 21]. Respiratory muscles seem to exhibit similar adaptations to training to those of other skeletal muscles provided the training follows the basic training principals for any other striated muscles with regard to intensity, duration of the stimulus and the specifics of the trainings [17, 21]. Current consensus statements confine recommendation of inspiratory muscle training to patients with inspiratory muscle weakness [1, 13, 15, 32, 34]. Furthermore, there are a few randomised, controlled trials in which respiratory muscle training has elicited a positive effect upon neuromuscular diseases [15, 32] as well as less rigorously designed and executed studies in conditions such as kyphoscoliosis [10]. Aldrich and Uhrlass [1] have shown that even in severe respiratory failure, inspiratory muscles can be trained successfully in patients with DMD.
In the current study, %FVC increased in all patients. Although %FVC is usually used as a parameter for the progression of the neuromuscular disease involving the entire respiratory system, it is affected by respiratory muscle function [6, 17]. Fry et al. [9] found inspiratory muscle training significantly increased inspiratory muscle strength in patients with neuromuscular disease and there was also a clinically important positive effect on expiratory pulmonary function measures (%FVC, FEV1, FEF25–75%) substantiating the indirect positive effects of the training on expiratory pulmonary function. Koessler et al. [17] have shown that inspiratory muscle training improves inspiratory muscle strength and endurance and that the training leads to a stabilization of %FVC even in the long-term of 2 years. In our series, %FVC increased after inspiratory muscle training for 6 weeks prior to surgery. There was significant difference between the values before and after training (22 vs. 26%). Although the number of our patients is relatively small, the efficacy of preoperative inspiratory muscle training has been shown clinically in DMD patients with severely compromised pulmonary function in this study.
In the current study, there was no postoperative pulmonary complication although our patients had a mean FVC of 22% of predicted value. It has been well recognized that DMD patients are at high-risk for pulmonary complications following surgery [2, 8, 10, 11, 22]. A positive correlation between decreased %FVC and increased postoperative pulmonary complications in children with neuromuscular scoliosis undergoing spinal fusion has been reported [27]. The reported postoperative complications included temporary or permanent tracheotomy, respiratory infection and ventilator dependency [22]. These complications are associated with decreased expiratory pulmonary function in this group of patients. Fry et al. [9] showed inspiratory muscle training increased inspiratory muscle strength and resulted in generalized improvements in expiratory pulmonary function in patients with neuromuscular disease. Although expiratory pulmonary function was not measured in our series, we suggest it should have been improved after inspiratory muscle training and thereby there was no postoperative pulmonary complication. The other reason for the excellent results is the multi-disciplinary team involved with the patients’ care. All the operations were performed by one well experienced spine surgeon. The nursing and physiotherapy staff were also well experienced. All the patients were extubated after confirmation of spontaneous breathing in the operating room and thereby they did not become ventilator dependent. They were encouraged to sit up in bed on the operative day and frequent expulsion of debris was performed by nursing staffs in the immediately postoperative period. However, if respiratory complications occur, noninvasive ventilatory support (e.g. IPPV or MAC), or invasive ventilatory support (e.g. reintubation or tracheotomy) should be considered.
For such patients with respiratory muscle weakness associated with DMD, sudden decreases in SpO2, resulting in respiratory failure, may occur in the immediate postoperative period. This condition should be assumed to be due to air way mucous encumberment. Bach et al. [3] recommended the use of mechanically assisted coughing (MAC) to reverse this condition. MAC is a device that provides a deep lung insufflation immediately followed by a forced exsufflation via oranasal mask, to rapidly empty the airways of debris. This device has been used for patients with neuromuscular disease to increase cough flows to clear airway secretions during intercurrent respiratory tract infections, thereby they can avoid pneumonias and respiratory failure.
Although longer-term follow-up must be performed to determine the final fate of our series, this study shows that our patients with high-risk pulmonary dysfunction and severe scoliosis in DMD could undergo surgical correction of scoliosis with general anesthesia after inspiratory muscle training. %FVC increased in all patients after training. All patients were extubated on the operative day. There was a smooth transition to spontaneous breathing, reducing the ICU stay. Curve correction and maintenance of correction was excellent. There were no major complications. No continuous or prolonged ventilatory support was needed in the immediately postoperative period. Severe scoliosis with poor pulmonary function in DMD patients has been considered a reason to avoid surgery mainly because of the fear of peri/postoperative respiratory complications. However, we feel such patients with FVC considered too low to permit reasonable surgical risk can undergo surgery with general anesthesia using the described method and can benefit from posterior spinal fusion for scoliosis.
References
Aldrich TK, Uhrlass RM (1987) Weaning from mechanical ventilation: successful use of modified inspiratory resistive training in muscular dystrophy. Crit Care Med 15:247–249
Askin GN, Hallettv R, Hare N et al (1997) The outcome of scoliosis surgery in the severely handicapped child: an objective and subjective assessment. Spine 22:44–50
Bach JR, Sabharwal S (2005) High pulmonary risk scoliosis surgery. J Spinal Disord Tech 18:527–530
Bell DF, Moseley CF, Koneska J (1989) Unit rod segmental instrumentation in the management of patients with progressive neuromuscular spinal deformity. Spine 14:1301–1307
Bonnett C, Brown JC, Perry J et al (1995) Evolution of treatment of paralytic scoliosis at Ranchos Los Amigos Hospital. J Bone Joint Surg 57A:206–215
Braun N, Narinder SA, Rochester DF (1983) Respiratory muscles and pulmonary function in polyositis and other proximal myopathies. Thorax 38:616–623
Bridwell KH, Baldus C, Iffrig TM et al (1999) Process measures and patient/parent evaluation of surgical management of spinal deformities in patients with progressive flaccid neuromuscular scoliosis (Duchenne’s muscular dystrophy and spinal muscular atrophy). Spine 24:1300–1309
Dias RC, Miller F, Dabney K et al (1996) Surgical correction of spinal deformity using a unit rod in children with cerebral palsy. J Pediatr Orhop 16:734–740
Fry DK, Pfalzer LA, Chokshi AR, Wagner MT, Jackson ES (2007) Randomized control trial of effects of a 10-week inspiratory muscle training program on measures of pulmonary function in persons with multiple sclerosis. J Neurol Phys Therapy 31:162–172
Galasko CBS, Delany C, Morris P (1992) Spinal stabilization in Duchenne muscular dystrophy. J Bone Joint Surg 74B:210–220
Gill I, Eagle M, Mehta JS, Gibson MJ (2006) Correction of neuromuscular scoliosis in patients with preexisting respiratory failure. Spine 31:2478–2483
Hornstein S, Inman S, Ledsome JR (1987) Ventilatory muscle training in kyphoscoliosis. Spine 12:859–863
Hsu JD (1983) The natural history of spinal curve progression in the nonambulatory Duchenne muscular dystrophy patient. Spine 8:771–775
Jenkins JG, Bohn D, Edmonds JF et al (1982) Evaluation of pulmonary function in muscular dystrophy patients requiring spinal surgery. Crit Care Med 10:645–648
Kennedy JD, Staples AJ, Brook PD et al (1995) Effect of spinal surgery on lung function in Duchenne muscular dystrophy. Thorax 50:1173–1178
Klefbeck B, Nedjad JH (2003) Effect of inspiratory muscle training in patients with multiple sclerosis. Arch Phys Med Rehabil 84:994–999
Koessler W, Wanke T, Winkler G et al (2001) 2 years’ experience with inspiratory muscle training in patients with neuromuscular disorders. Chest 120:765–769
Kurz LT, Mubarak SJ, Schultz P et al (1983) Correlation of scoliosis and pulmonary function in Duchenne muscular dystrophy. J Paediatr Orthop 3:347–355
Larsson EL, Aaro S, Oberg B (1999) Activities and functional assessment 1 year after spinal fusion for paralytic scoliosis. Eur Spine J 8:100–109
Leith DE, Bradley M (1976) Ventilatory muscle strength and endurance training. J Appl Physiol 41:508–516
Macklem PT (1986) Muscular weakness and respiratory function. N Engl J Med 314:775–776
Marsh A, Edge G, Lehovsky J (2003) Spinal fusion in patients with Duchenne’s muscular dystrophy and a low forced vital capacity. Euro Spine J 12:507–512
McDonald CM, Abresch RT, Carter GT et al (1995) Profiles of neuromuscular disease: Duchenne muscular dystrophy. Am J Phys Med Rehabil 74(Suppl):70–92
Miller RG, Chalmers AC, Dao H, Filler-Katz A (1991) The effects of spine fusion on respiratory function in Duchenne muscular dystrophy. Neurology 41:38–45
Mubarak SJ, Morin WD, Leach J (1993) Spinal fusion in Duchenne muscular dystrophy and fusion to the sacro-pelvis? J Paediatr Orthop 13:752–760
Oda T, Shimizu N, Yonenobu K et al (1993) Longitudinal study of spinal deformity in Duchenne muscular dystrophy. J Paediatr Orthop 13:478–488
Padman R, McNamara R (1990) Postoperative pulmonary complications in children neuromuscular scoliosis who underwent posterior spinal fusion. Del Med J 62:999–1003
Rawlins BA, Winter RB, Lonstein JF et al (1996) Reconstructive spinal surgery in pediatric patients with major loss in vital capacity. J Pediatr Orthop 16:284–292
Rideau Y, Glorinon B, Delaubier A et al (1984) The treatment of scoliosis in Duchenne muscular dystrophy. Muscle Nerve 7:281–286
Sakai DN, Hsu JD, Bonnett DA et al (1997) Stabilization of the collapsing spine in Duchenne muscular dystrophy. Clin Orthop 128:256–260
Shapiro F, Specht L (1993) The diagnosis and orthopaedic treatment of inherited muscular diseases of childhood. J Bone Joint Surg 75A:439–445
Smith A, Koreska J, Mosley CF (1989) Progression of scoliosis in Duchenne muscular dystrophy. J Bone Joint Surg 71A:1066–1074
Sticker U, Moser H, Aebi M (1996) Predominantly posterior instrumentation and fusion in neuromuscular and neurogenic scoliosis in children and adolescents. Euro Spine J 5:101–106
Sullivan JA, Conner SB (1982) Comparison of Harrington instrumentation and segmental spinal instrumentation in the management of neuromuscular spinal deformity. Spine 7:299–304
Sussman MD (1984) Advantage of early spinal stabilization and fusion in patients with Duchenne muscular dystrophy. J Paediatr Orthop 4:532–538
Swank SM, Brown JC, Perry RE (1982) Spinal fusion in Duchenne’s muscular dystrophy. Spine 7:484–491
Conflict of interest statement
No funds were received in support of this study. The author(s) of this manuscript has/have chosen not to furnish Ejost and its readers with information regarding any relationship that might exist between a commercial party and material contained in this manuscript that might represent a potential conflict of interest. No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takaso, M., Nakazawa, T., Imura, T. et al. Preoperative inspiratory muscle training for patients with severe scoliosis and high-risk pulmonary dysfunction in duchenne muscular dystrophy. Eur J Orthop Surg Traumatol 20, 113–121 (2010). https://doi.org/10.1007/s00590-009-0500-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00590-009-0500-4