Article Text
Statistics from Altmetric.com
A review of the newly published guidelines on the management of pleural infection in children
Commissioned by the British Thoracic Society (BTS) Standards of Care Committee, the BTS guidelines for the management of pleural infection in children are published as a supplement to this month’s issue of Thorax.1 They have also been subject to scrutiny by the Quality of Practice Committee (QPC) of the Royal College of Paediatrics and Child Health (RCPCH) using the AGREE appraisal instrument, and consequently have been endorsed by the RCPCH. So why did we write them and was it worth it? More importantly—should you read them? What follows are my views as chair of the guideline committee and may not represent those of the other members of the committee (listed in the Acknowledgements).
WHY DID WE WRITE THEM?
We are not paediatricians for nothing—we understand sibling rivalry—so when the adult respiratory physicians got an empyema guideline,2 we wanted one. However, it was more than just that. There are major differences between adult and paediatric pleural infections. The principal one is that most children are fit and well before they contract pneumonia and develop an empyema, so the outcome is almost always excellent. This is in contrast to the disease in adults where empyema is a cause of significant morbidity and mortality (recent data has revealed a 12% mortality rate even in those without cancer3). Nevertheless, management of childhood empyema can still be a therapeutic challenge. Unfortunately, in the UK there is little consensus over best practice, both among respiratory paediatricians and also thoracic surgeons. It was therefore considered important to synthesise the available evidence and write guidelines, even though they may only point people in the right direction and highlight the unresolved issues. It is hoped that the guidelines will facilitate dissemination of evidence and standardisation of patient care. While they are principally aimed at the UK, they are applicable to children worldwide. The principal differences in management from the UK will concern predominant local pathogens, availability of antibiotics and fibrinolytics, and availability of trained personnel.
HOW DID WE WRITE THEM?
For anyone unfamiliar with the process of writing a guideline, the website of the Scottish Intercollegiate Guideline Network (SIGN) is invaluable (www.sign.ac.uk) and we followed their process for producing levels of evidence and grades of recommendations. Literature searching was extensive and professional (library of National Heart Lung Institute) and the search strategy is included as an appendix to the guidelines. It was kept up to date and rechecked by individual members responsible for a particular topic. We ensured the make-up of the guideline committee included all the relevant disciplines: a general/emergency paediatrician, respiratory paediatricians (including a trainee), a paediatric and thoracic surgeon, a microbiologist and a radiologist. We also ensured that representatives of the different major viewpoints were included (readily identifiable from previous pro/con debates). With the use of email, only one meeting was required with the whole group. Where there was no evidence we relied on “expert opinion” and, while we did not use a formal consensus technique, the guidelines are a fair reflection of the opinions of the whole committee as well as the specialist reviewers. It was pointed out by the RCPCH that we did not have “consumer input”. While I would agree that guidelines on chronic diseases such as asthma or cystic fibrosis benefit from patient/parent input, I do not believe this to be case with a one-off acute condition such as an empyema where parents will not have gained a particular expertise. The guidelines went through several review stages including the BTS Standards of Care Committee, specialist reviewers (listed below), and the RCPCH QPC. Our thanks are due to them all for their valuable contributions.
WHAT PROBLEMS DID WE ENCOUNTER?
Guidelines need to be evidence-based, so this immediately presented a problem as there is a lack of evidence from randomised controlled trials for almost all the relevant issues in childhood empyema. In addition, it is inappropriate simply to extrapolate adult data to children, particularly when the condition behaves so differently in the two populations. Why so few trials? Empyema has until recently been relatively rare in developed countries. Over the last decade there has been a noticeable increase both in the UK and the USA;1 in our own institution 20 cases were reported during the 5 year period from 1990 to 1994 inclusive,4 whereas we now see that many cases each year. While this may reflect a change in referral patterns, undoubtedly there has been a genuine increase in incidence. In the UK the predominant pathogen is penicillin-sensitive pneumococcus, while in developing countries it is still Staphylococcus aureus.1 It is possible that the increased incidence in the UK coincides with a reduction in antibiotic prescribing in primary care.5 Although it is good that less amoxicillin is given to “chesty” children with viral upper respiratory tract infections, the cost may be a missed opportunity for early treatment of the few children with genuine pneumonia.
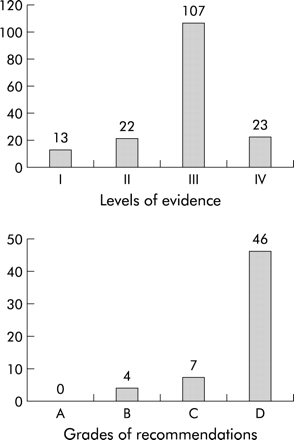
As management of empyema became more of an issue, the British Paediatric Respiratory Society Empyema Study Group (led by Anne Thomson in Oxford) performed the only major placebo controlled randomised trial in childhood empyema—a multicentre trial of intrapleural urokinase that still took two winters to enrol 60 cases.6 This trial provided three of the four SIGN grade B recommendations in the guidelines. The only other grade B recommendation was related to using microbiology results to guide antibiotic choice (grade taken from adult guidelines), but even then the RCPCH QPC downgraded that to a D. There was not a single grade A recommendation, seven grade Cs, and 46 grade Ds (fig 1). This means that 80% of our recommendations were based on evidence taken from case reports, case series, and expert opinion. This should not be used as a criticism of the guidelines; it is simply a reflection of the available evidence. In addition, many of the grade D recommendations are based on safe current practice and common sense. No one can deny that “analgesia is important to keep the child comfortable, particularly in the presence of a chest drain”, a grade D recommendation that never has been (nor ever should be) subjected to a randomised controlled trial. Perhaps SIGN should come up with an extra grade (E)—no evidence but blindingly obvious. Our grade D recommendation that “all children with parapneumonic effusion or empyema should be admitted to hospital” would fit that category. Evidence from randomised controlled trials should not be the only resource that informs guidelines, and clinical experience should not be excluded.
Levels of evidence and grades of recommendation using SIGN ratings.1
WHAT DID WE RECOMMEND?
There are 57 bulleted recommendations with accompanying explanatory text and one algorithm summarising management. The guidelines need reading in full, but there are a few points worth highlighting. There is no role for routine lateral chest radiographs or CT scans; plain chest radiographs and ultrasound are sufficient and will reduce the radiation burden. Pleural fluid must be sent for cell differential counting (or cytology) as well as microbiological analysis, but there is nothing to gain from biochemical analysis (such as pH, glucose, protein, lactate dehydrogenase). If there is any suspicion of malignancy, a small volume diagnostic tap for cytological testing is needed, where possible avoiding general anaesthesia/sedation to prevent potentially disastrous superior mediastinal obstruction. Significant effusions should be drained from the outset; repeated taps are not recommended.
We recommend that a respiratory paediatrician should be involved early in the care of all patients requiring chest tube drainage for an empyema. We feel strongly that this condition should be looked after in tertiary centres, not least because of the nursing expertise required to safely manage chest drains. Empyema is still relatively uncommon and, given the uncertainties over management, we feel it is not ideal for patients to be treated in a general paediatric unit that might only treat one or two cases a year. To an extent this recommendation for referral is happening anyway, partly because constraints imposed by junior doctor working hours and the European Working Time Directive mean that many paediatric specialist registrars are no longer experienced at chest drain insertion. We also find that many anaesthetists in district general hospitals are now unwilling to anaesthetise children with respiratory compromise. Interestingly, this recommendation—which I believe to be one of the most important in the whole guideline—was the only one that the RCPCH QPC was not keen on publishing in the version due to be sent to all UK paediatricians. Clearly, there will be circumstances when it is unnecessary or not in the child’s best interest to move large distances, but there is a concern that medicolegal consequences may follow if management goes wrong outside a tertiary centre.
We have suggested some antibiotic regimens but local antibiotic policies and likely pathogens should also be taken into account. There is no evidence that large bore drains confer any advantage over small ones, but they certainly cause more pain and reduce mobility, so small ones (8–12 FG) should be used where possible. They should be placed at the optimum site suggested by ultrasound, but not too far posteriorly as they are uncomfortable to lie on and there is a greater risk of traumatic bleeding from the posterior intercostal spaces. One of the more contentious issues was whether drains should be inserted under general anaesthesia or sedation with local anaesthesia. Most anaesthetists would not endorse intravenous sedation (especially in a child with respiratory compromise), but a definitive anaesthetic guideline does not exist to clarify this issue. Personally, I believe a general anaesthetic is kinder for the child (especially younger ones); it also makes the procedure easier and allows time for a long line to be inserted. The compromise for the guidelines was our recommendation that intravenous sedation should only be given by those trained in the use of conscious sedation, airway management and resuscitation of children, using full monitoring equipment—something which I suspect will rule out many paediatricians anyway.
For those having the medical option (see algorithm of guideline), based on a randomised controlled trial,6 use of intrapleural urokinase is recommended when there is thick fluid with loculations or overt pus. The biggest issue is the role of surgery as primary treatment. There is little dispute that failure of chest tube drainage, antibiotics, and fibrinolytic agents should prompt discussion with a thoracic surgeon, and that patients should be considered for surgical treatment if they have persisting sepsis in association with a persistent pleural collection. The issue where consensus cannot be reached in the UK is the role of medical versus early surgery and, if surgery is chosen, whether a mini-thoracotomy or video assisted thoracoscopic surgery (VATS) is superior. There are no trial data to inform the decision; however, there are many case series, mostly written by enthusiasts of their own technique, using historical “controls”. For this reason our guideline gives medical and surgical options in the management algorithm. For now, units should continue to manage their cases using their own established and familiar practices, in whatever way they feel is best from their own experiences. Available resources may also affect local policy, especially in health services with financial constraints. Parents and children need to be fully informed of the options (good patient information sheets are essential), but ultimately they are likely to be swayed by the opinion of the treating doctors and nurses. In the case series of 20 patients treated at the Royal Brompton Hospital during 1990–4, 65% had immediate surgery but 90% eventually required surgery;4 currently, with the use of small drains and urokinase, only 5–10% require surgery in our centre. What is needed is an understanding of the factors that predict failure of medical management, as these children could then be selected for immediate surgery while the rest could have simple medical management.
FUTURE RESEARCH
The guidelines offer a list of potential areas for future clinical research. In terms of management, the issue of drain/fibrinolytics versus early mini-thoracotomy versus VATS is the outstanding one. Since the outcome is generally so good in terms of clinical recovery, lung function and chest radiographic appearance (whatever the treatment), it will be difficult to prove one better than another, and most treatment regimens will show equivalence. Pain and discomfort must be taken into account, not just days in hospital, as most children would prefer an extra day or so in hospital if it meant significantly less pain. It is likely that large multicentre trials will be necessary to make any serious progress, although there is an ongoing study at Great Ormond Street Hospital for Children comparing intrapleural urokinase with early VATS in a randomised non-blinded trial.
CONCLUSIONS
I will conclude by quoting a statement from the RCPCH guideline appraisal: “Guidelines are not intended to restrict clinical freedom, but practitioners are expected to use the recommendations as a basis for their practice. Local resources and the circumstances and preferences of individual patients will need to be taken into account”. This is certainly true of childhood empyema and, yes, these BTS guidelines should be read.
Acknowledgments
Paediatric Pleural Diseases Subcommittee of the BTS Standards of Care Committee: Dr I M Balfour-Lynn (Chair), Dr E Abrahamson, Mr G Cohen, Dr J Hartley, Dr S King, Mr D Parikh, Dr D Spencer, Dr A H Thomson, Dr D Urquhart. Also Dr B G Higgins (past Chair), Dr D Boldy (present Chair) and the other members of the BTS Standards of Care Committee.
Specialist reviewers: Dr R Dinwiddie, Dr I Doull, Mr P Goldstraw, Dr R A Primhak, Dr P Seddon. Also Dr H Baumer (Chair) and other members of RCPCH Quality of Practice Committee.
A review of the newly published guidelines on the management of pleural infection in children
Linked Articles
- airwaves